stdClass Object
(
[nazev] => Department of Organic Chemistry
[adresa_url] =>
[api_hash] =>
[seo_desc] =>
[jazyk] =>
[jednojazycny] =>
[barva] =>
[indexace] => 1
[obrazek] =>
[ga_force] =>
[cookie_force] =>
[secureredirect] =>
[google_verification] => UOa3DCAUaJJ2C3MuUhI9eR1T9ZNzenZfHPQN4wupOE8
[ga_account] => UA-10822215-3
[ga_domain] =>
[ga4_account] => G-VKDBFLKL51
[gtm_id] =>
[gt_code] =>
[kontrola_pred] =>
[omezeni] => 0
[pozadi1] =>
[pozadi2] =>
[pozadi3] =>
[pozadi4] =>
[pozadi5] =>
[robots] =>
[htmlheaders] =>
[newurl_domain] => 'uoch.vscht.cz'
[newurl_jazyk] => 'en'
[newurl_akce] => '[en]'
[newurl_iduzel] =>
[newurl_path] => 8548/6214/6521
[newurl_path_link] => Odkaz na newurlCMS
[iduzel] => 6521
[platne_od] => 31.10.2023 17:15:00
[zmeneno_cas] => 31.10.2023 17:15:57.546654
[zmeneno_uzivatel_jmeno] => Jan Kříž
[canonical_url] =>
[idvazba] => 7334
[cms_time] => 1713563497
[skupina_www] => Array
(
)
[slovnik] => stdClass Object
(
[logo_href] => /
[logo] =>  [google_search] => 001523547858480163194:u-cbn29rzve
[social_fb_odkaz] => https://www.facebook.com/uoch.vscht
[social_tw_odkaz] => https://twitter.com/uoch_vscht
[social_yt_odkaz] =>
[paticka_budova_a_nadpis] => BUILDING A
[paticka_budova_a_popis] => Rector,
Department of Communications,
Department of Education,
FCT Dean’s Office,
Centre for Information Services
[paticka_budova_b_nadpis] => BUILDING B
[paticka_budova_b_popis] => Department of R&D, Dean’s Offices:
FET,
FFBT,
FCE,
Computer Centre,
Department of International Relations,
Bursar
[paticka_budova_c_nadpis] => BUILDING C
[paticka_budova_c_popis] =>
Crèche Zkumavka,
General Practitioner,
Department of Economics and Management,
Department of Mathematics
[paticka_budova_1_nadpis] => NATIONAL LIBRARY OF TECHNOLOGY
[paticka_budova_1_popis] =>
[paticka_budova_2_nadpis] => CAFÉ CARBON
[paticka_budova_2_popis] =>
[paticka_adresa] => UCT Prague
[google_search] => 001523547858480163194:u-cbn29rzve
[social_fb_odkaz] => https://www.facebook.com/uoch.vscht
[social_tw_odkaz] => https://twitter.com/uoch_vscht
[social_yt_odkaz] =>
[paticka_budova_a_nadpis] => BUILDING A
[paticka_budova_a_popis] => Rector,
Department of Communications,
Department of Education,
FCT Dean’s Office,
Centre for Information Services
[paticka_budova_b_nadpis] => BUILDING B
[paticka_budova_b_popis] => Department of R&D, Dean’s Offices:
FET,
FFBT,
FCE,
Computer Centre,
Department of International Relations,
Bursar
[paticka_budova_c_nadpis] => BUILDING C
[paticka_budova_c_popis] =>
Crèche Zkumavka,
General Practitioner,
Department of Economics and Management,
Department of Mathematics
[paticka_budova_1_nadpis] => NATIONAL LIBRARY OF TECHNOLOGY
[paticka_budova_1_popis] =>
[paticka_budova_2_nadpis] => CAFÉ CARBON
[paticka_budova_2_popis] =>
[paticka_adresa] => UCT Prague
Technická 5
166 28 Prague 6 – Dejvice
IČO: 60461373 / VAT: CZ60461373
Czech Post certified digital mail code: sp4j9ch
Copyright: UCT Prague 2015
Information provided by the Department of Organic Chemistry. Technical support by the Computing Centre.
[paticka_odkaz_mail] => mailto:kundrato@vscht.cz
[social_fb_title] => Facebook of Department
[social_tw_title] => Twitter of Department
[social_yt_title] =>
[aktualizovano] => Updated
[autor] => Author
[paticka_mapa_alt] =>
[drobecky] => You are here: VŠCHT Praha – FCHT – ÚOCH
[intranet_odkaz] => http://intranet.vscht.cz/
[intranet_text] => Intranet
[logo_mobile_href] => /
[logo_mobile] =>  [mobile_over_nadpis_menu] => Menu
[mobile_over_nadpis_search] => Search
[mobile_over_nadpis_jazyky] => Languages
[mobile_over_nadpis_login] => Login
[menu_home] => Homepage
[zobraz_desktop_verzi] => switch to desktop version
[zobraz_mobilni_verzi] => switch to mobile version
[more_info] => more information
[paticka_mapa_odkaz] =>
[nepodporovany_prohlizec] => For full access, please use different browser.
[preloader] => Wait a second...
[social_in_odkaz] =>
[den_kratky_4] =>
[archiv_novinek] =>
[novinky_servis_archiv_rok] =>
[novinky_kategorie_1] =>
[novinky_kategorie_2] =>
[novinky_kategorie_3] =>
[novinky_kategorie_4] =>
[novinky_kategorie_5] =>
[novinky_archiv_url] =>
[novinky_servis_nadpis] =>
[novinky_dalsi] =>
[hledani_nadpis] => hledání
[hledani_nenalezeno] => Nenalezeno...
[hledani_vyhledat_google] => vyhledat pomocí Google
[social_li_odkaz] =>
)
[poduzel] => stdClass Object
(
[6525] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[6532] => stdClass Object
(
[obsah] =>
[iduzel] => 6532
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6530] => stdClass Object
(
[obsah] =>
[iduzel] => 6530
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6531] => stdClass Object
(
[obsah] =>
[iduzel] => 6531
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
)
[iduzel] => 6525
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6526] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[21923] => stdClass Object
(
[nazev] => Department of Organic Chemistry
[seo_title] => Department of Organic Chemistry
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
[mobile_over_nadpis_menu] => Menu
[mobile_over_nadpis_search] => Search
[mobile_over_nadpis_jazyky] => Languages
[mobile_over_nadpis_login] => Login
[menu_home] => Homepage
[zobraz_desktop_verzi] => switch to desktop version
[zobraz_mobilni_verzi] => switch to mobile version
[more_info] => more information
[paticka_mapa_odkaz] =>
[nepodporovany_prohlizec] => For full access, please use different browser.
[preloader] => Wait a second...
[social_in_odkaz] =>
[den_kratky_4] =>
[archiv_novinek] =>
[novinky_servis_archiv_rok] =>
[novinky_kategorie_1] =>
[novinky_kategorie_2] =>
[novinky_kategorie_3] =>
[novinky_kategorie_4] =>
[novinky_kategorie_5] =>
[novinky_archiv_url] =>
[novinky_servis_nadpis] =>
[novinky_dalsi] =>
[hledani_nadpis] => hledání
[hledani_nenalezeno] => Nenalezeno...
[hledani_vyhledat_google] => vyhledat pomocí Google
[social_li_odkaz] =>
)
[poduzel] => stdClass Object
(
[6525] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[6532] => stdClass Object
(
[obsah] =>
[iduzel] => 6532
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6530] => stdClass Object
(
[obsah] =>
[iduzel] => 6530
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6531] => stdClass Object
(
[obsah] =>
[iduzel] => 6531
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
)
[iduzel] => 6525
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6526] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[21923] => stdClass Object
(
[nazev] => Department of Organic Chemistry
[seo_title] => Department of Organic Chemistry
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] => New PhD topics of our department: here
All publications of our authors (since 2015): here
[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[iduzel] => 21923
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /home
[sablona] => stdClass Object
(
[class] => boxy
[html] =>
[css] =>
[js] => $(function() {
setInterval(function () { $('*[data-countdown]').each(function() { CountDownIt('#'+$(this).attr("id")); }); },1000);
setInterval(function () { $('.homebox_slider:not(.stop)').each(function () { slide($(this),true); }); },5000);
});
function CountDownIt(selector) {
var el=$(selector);foo = new Date;
var unixtime = el.attr('data-countdown')*1-parseInt(foo.getTime() / 1000); if(unixtime<0) unixtime=0;
var dnu = 1*parseInt(unixtime / (3600*24)); unixtime=unixtime-(dnu*(3600*24));
var hodin = 1*parseInt(unixtime / (3600)); unixtime=unixtime-(hodin*(3600));
var minut = 1*parseInt(unixtime / (60)); unixtime=unixtime-(minut*(60));
if(unixtime<10) {unixtime='0'+unixtime;}
if(dnu<10) {unixtime='0'+dnu;}
if(hodin<10) {unixtime='0'+hodin;}
if(minut<10) {unixtime='0'+minut;}
el.html(dnu+':'+hodin+':'+minut+':'+unixtime);
}
function slide(el,vlevo) {
if(el.length<1) return false; var leva=el.find('.content').position().left; var sirka=el.width(); var pocet=el.find('.content .homebox').length-1;
var cislo=leva/sirka*-1; if(vlevo) { if(cislo+1>pocet) cislo=0; else cislo++; } else { if(cislo==0) cislo=pocet-1; else cislo--; }
el.find('.content').animate({'left':-1*cislo*sirka});
el.find('.slider_puntiky a').removeClass('selected');
el.find('.slider_puntiky a.puntik'+cislo).addClass('selected');
return false;
}
function slideTo(el,cislo) {
if(el.length<1) return false; var sirka=el.width(); var pocet=el.find('.content .homebox').length-1; if(cislo<0 || cislo>pocet) return false;
el.find('.content').animate({'left':-1*cislo*sirka});
el.find('.slider_puntiky a').removeClass('selected');
el.find('.slider_puntiky a.puntik'+cislo).addClass('selected');
return false;
}
[autonomni] => 1
)
)
[16022] => stdClass Object
(
[nazev] => Department of Organic Chemistry - Staff
[seo_title] => Department of Organic Chemistry
[seo_desc] =>
[autor] =>
[autor_email] =>
[perex] =>
[ikona] => telefon-zvoni
[obrazek] =>
[ogobrazek] =>
[pozadi] =>
[obsah] =>
| k Phonebook of the Department | |||
Professors: |
|||
|
Prof. Radek Cibulka e +420 22044 4182 d A 278c |
Head of Department | www | ResearcherID |
|
Prof. Pavel Lhoták e +420 22044 5055 d A 249 |
www | ||
|
Prof. Jaroslav Kvíčala e +420 22044 4240 d A 278d |
www | ||
|
Prof. Jiří Svoboda e +420 22044 3688 d A 278e |
www | ||
Associate Professors: |
|||
|
Assoc. Prof. Jan Budka e +420 22044 4284 d A 251 |
DOC web editor | www | |
|
Assoc. Prof. Jana Hodačová e +420 22044 4173 d A 270 |
www | ||
|
Assoc. Prof. Michal Kohout e +420 22044 5012 d A 258 |
www | ||
|
Assoc. Prof. Igor Linhart e +420 22044 4165 d A 266 |
www | ||
|
Assoc. Prof. Tomáš Tobrman e +420 22044 4245 d A 271 |
RIT | www | |
Assistant Professors: |
|||
|
Dr. Michal Himl e +420 22044 4165 d A 266 |
www | ||
|
Dr. Roman Holakovský e +420 22044 4279 d A 255 |
www | ||
|
Dr. Petr Kovaříček e +420 22044 2040 d B3 207 |
www | ||
|
Dr. Václav Kozmík e +420 22044 4118 d A 308 |
www | ||
|
Dr. Martin Krupička e +420 220 444 173 |
www | ||
|
Dr. Ondřej Kundrát e +420 22044 4280 d A 253 |
www | ResearcherID | |
|
Dr. Petra Ménová e +420 22044 3686 d A 268 |
www | ||
|
Dr. Pavla Perlíková e +420 22044 2039 d B3 206 |
www | ||
|
Dr. Markéta Rybáčková e +420 22044 4242 d A 259 |
Secretary of Department |
www | |
|
Dr. Eva Svobodová e +420 22044 4249 d A 260 |
www | ||
Technicians: |
|||
|
Květa Bártová e +420 22044 3686 d A 268 |
|||
|
Ivana Bocková, MSc. e +420 22044 4280 d A 250 |
|||
| Michaela Kadlecová
e +420 22044 4276 d A 262 b Michaela.Kadlecova@vscht.cz |
|||
|
Martina Kovandová e +420 22044 4279 d A 255 |
|||
|
Vladimír Kuneš e +420 22044 4277 d A L06 |
|||
|
Jana Netušilová e +420 22044 4164 d A 278a |
Administrative Support of Department | ||
|
Markéta Slabochová, MSc. e +420 22044 4059 d A 308 |
Economist of Department | ||
|
Helena Štenglová e +420 22044 4278 d A 257 |
|||
|
Ing. David Tetour e +420 22044 4173 d A270 |
|||
|
Marta Tokárová e +420 220 444 245 d A 271 |
|||
Essential Information for Current Students
Bachelor Students
Master Students
PhD Students
Lectures delivered by members of the Department
- Toxicology and ecology
- Organic chemistry I, II and III
- Structural analysis
- Reactivity of organic compounds
- Molecular design
- Organic synthesis I and II
- Quantum organic chemistry
- Farmacochemistry
- Organic stereochemistry
- Organometallic chemistry
- Organic chemistry of selected elements
DATA
stdClass Object
(
[nazev] => Department of Organic Chemistry - Staff
[seo_title] => Department of Organic Chemistry
[seo_desc] =>
[autor] =>
[autor_email] =>
[perex] =>
[ikona] => telefon-zvoni
[obrazek] =>
[ogobrazek] =>
[pozadi] =>
[obsah] =>
| k Phonebook of the Department | |||
Professors: |
|||
|
Prof. Radek Cibulka e +420 22044 4182 d A 278c |
Head of Department | www | ResearcherID |
|
Prof. Pavel Lhoták e +420 22044 5055 d A 249 |
www | ||
|
Prof. Jaroslav Kvíčala e +420 22044 4240 d A 278d |
www | ||
|
Prof. Jiří Svoboda e +420 22044 3688 d A 278e |
www | ||
Associate Professors: |
|||
|
Assoc. Prof. Jan Budka e +420 22044 4284 d A 251 |
DOC web editor | www | |
|
Assoc. Prof. Jana Hodačová e +420 22044 4173 d A 270 |
www | ||
|
Assoc. Prof. Michal Kohout e +420 22044 5012 d A 258 |
www | ||
|
Assoc. Prof. Igor Linhart e +420 22044 4165 d A 266 |
www | ||
|
Assoc. Prof. Tomáš Tobrman e +420 22044 4245 d A 271 |
RIT | www | |
Assistant Professors: |
|||
|
Dr. Michal Himl e +420 22044 4165 d A 266 |
www | ||
|
Dr. Roman Holakovský e +420 22044 4279 d A 255 |
www | ||
|
Dr. Petr Kovaříček e +420 22044 2040 d B3 207 |
www | ||
|
Dr. Václav Kozmík e +420 22044 4118 d A 308 |
www | ||
|
Dr. Martin Krupička e +420 220 444 173 |
www | ||
|
Dr. Ondřej Kundrát e +420 22044 4280 d A 253 |
www | ResearcherID | |
|
Dr. Petra Ménová e +420 22044 3686 d A 268 |
www | ||
|
Dr. Pavla Perlíková e +420 22044 2039 d B3 206 |
www | ||
|
Dr. Markéta Rybáčková e +420 22044 4242 d A 259 |
Secretary of Department |
www | |
|
Dr. Eva Svobodová e +420 22044 4249 d A 260 |
www | ||
Technicians: |
|||
|
Květa Bártová e +420 22044 3686 d A 268 |
|||
|
Ivana Bocková, MSc. e +420 22044 4280 d A 250 |
|||
| Michaela Kadlecová
e +420 22044 4276 d A 262 b Michaela.Kadlecova@vscht.cz |
|||
|
Martina Kovandová e +420 22044 4279 d A 255 |
|||
|
Vladimír Kuneš e +420 22044 4277 d A L06 |
|||
|
Jana Netušilová e +420 22044 4164 d A 278a |
Administrative Support of Department | ||
|
Markéta Slabochová, MSc. e +420 22044 4059 d A 308 |
Economist of Department | ||
|
Helena Štenglová e +420 22044 4278 d A 257 |
|||
|
Ing. David Tetour e +420 22044 4173 d A270 |
|||
|
Marta Tokárová e +420 220 444 245 d A 271 |
|||
Publications of our authors:
2024
- Weisheitelová I., Varma N., Chudoba J., Burdziński G., Sikorski M., Cibulka R.:
Catalyst-free aerobic photooxidation of sensitive benzylic alcohols with chemoselectivity controlled by DMSO solvent.
Green Chem. 2024, accepted, https://doi.org/10.1039/D4GC00087K - Burešová Z., Gobeze H. B., Grygarová M., Pytela O., Klikar M., Obertík R., Cibulka R., Islam T., Schanze K. S., Bureš F.:
Dicyanopyrazine Photoredox Catalysts: Correlation of Efficiency with Photophysics and Electronic Structure.
J. Catal. 2024, 430, 115348, DOI: 10.1016/j.jcat.2024.115348 - Salvadori, K.; Churý, M.; Budka, J.; Harvalík, J.; Matějka, P.; Šimková, L.; Lhoták, P.:
Chemoselective Electrochemical Cleavage of Sulfonimides as a Direct Way to Sulfonamides.
J. Org. Chem. 2024, 89, 1425–1437, https://doi.org/10.1021/acs.joc.3c01932
2023
- Zubova, E., Pokluda A., Dvořáková, H., Krupička M., Cibulka R.:
Exploring the Reactivity of Flavins with Nucleophiles Using a Theoretical and Experimental Approach.
ChemPlusChem 2023, Accepted. DOI:10.1002/cplu.202300547. - Pavlovska T., Weisheitelová I., Pramthaisong C., Sikorski M., Jahn U., Cibulka R.:
Primary and Secondary Amines by Flavin‐Photocatalyzed Consecutive Desulfonylation and Dealkylation of Sulfonamides.
Adv. Synth. Catal. 2023, 365, 4662-4671. DOI:10.1002/adsc.202300843. - Golczak A., Prukała D., Gierszewski M., Cherkas V., Kwiatek D., Kubiak A., Varma N., Pedziński T., Murphree S., Cibulka R., Mrówczińska L., Kolanovski J. L., Sikorski M.:
Tetramethylalloxazines as efficient singlet oxygen photosensitizers and potential redox‑sensitive agents, Scientific Reports 2023, 13, 13426. DOI: 10.1038/s41598-023-40536-4.
' - Pokluda A., Zubova, E., Chudoba J., Krupička M., Cibulka R.:
Catalytic Artificial Nitroalkane Oxidases – a Way Towards Organocatalytic Umpolung.
Org. Biomol. Chem. 2023, 2768 – 2774. DOI: 10.1039/d3ob00101f. - Jurásek, B.; Fagan, P.; Dolenský, B.; Paškanová, N.; Dobšíková, K.; Raich, I.; Jurok, R.; Setnička, V.; Kohout, M.; Čejka, J.; Kuchař, M.:
A structural spectroscopic study of dissociative anaesthetic methoxphenidine. New J. Chem. 2023, 47, 4543.
https://doi.org/10.1039/d2nj06126k - Dobšíková, K.; Javorská, Ž.; Paškan, M.; Spálovská, D.; Trembuláková, P.; Herciková, J.; Kuchař, M.; Kozmík, V.; Kohout, M.; Setnička, V.:
Enantioseparation and a comprehensive spectroscopic analysis of novel synthetic cathinones laterally substituted with a trifluoromethyl group. Spectrochim. Acta, Part A, 2023, 291, 122320.
https://doi.org/10.1016/j.saa.2023.122320
2022
- Cortés F.R., Eigner V., Cuřínová P., Himl M.:
Structurally Forced Ion Binding Affinity: Tetraurea-Based Macrocycle as a Receptor for Ion Pairs
Eur. J. Org. Chem. 2022, e202200, DOI: 10.1002/ejoc.202200422
- Obertík R., Chudoba J., Šturala J., Tarábek J., Ludvíková L., Slanina T., König B., Cibulka R.:
Highly Chemoselective Catalytic Photooxidations Using Solvent as a Sacrificial Electron Acceptor
Chem. Eur. J. 2022, accepted as VIP. https://doi.org/10.1002/chem.202202487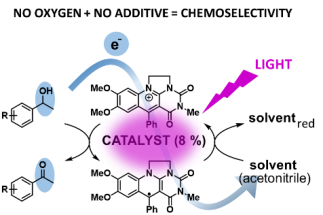
- Liška, A.; Řezanková, M.; Klíma, J.; Urban, J.; Budka, J.; Ludvík, J.:
Electrochemical, EPR, and quantum chemical study of reductive cleavage of cone-Calix[4]arene nosylates – New electrosynthetic approach
Electrochem. Sci. Adv. 2022, e2100221, DOI: org/10.1002/elsa.202100221 - Zhang, H.; Daněk, O.; Makarov, D.; Rádl, S.; Kim, D.; Ledvinka, J.; Vychodilová, K.; Hlaváč, J.; Lefèbre, J; Denis, M.; Rademacher, C. and Ménová, P.:
Drug-like Inhibitors of DC-SIGN Based on a Quinolone Scaffold
ACS Med. Chem. Lett. 2022, accepted. DOI: 10.1021/acsmedchemlett.2c00067 - Pavlovska T., Král Lesný D., Svobodová E., Hoskovcová I., Archipowa N., Kutta R. J., Cibulka R.:
Tuning Deazaflavins Towards Highly Potent Reducing Photocatalysts Guided by Mechanistic Understanding - Enhancement of the Key Step by the Internal Heavy Atom Effect. Chem. Eur. J. 2022, 28, e20220076. Hot Paper. DOI: 10.1002/chem.202200768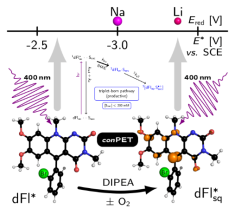
- Linhart, I.:
Toxikologie: Interakce škodlivých látek s živými organismy, jejich mechanismy, projevy a důsledky
VŠCHT Praha 2022 (3. vydání), ISBN 978-80-7592-103-1
- Linhart, I.; Himl, M.; Urban, V.; Mráz, J.:
Syntheses of methylcarbamoylated amino acids using synthetic equivalents of methyl isocyanate.
Synth. Commun. 2022, 52, 622-628. DOI: 10.1080/00397911.2022.2042563
2021
- Pokluda A., Anwar Z., Boguschová V., Anusiewicz I., Skurski P., Sikorski M., Cibulka R.:
Robust photocatalytic method using ethylene-bridged flavinium salts for the aerobic oxidation of unactivated benzylic substrates
Adv. Synth. Catal. 2021, 363, 4371-4379. VIP, DOI: 10.1002/adsc.202100024.
- Jakubec M., Novák D., Zatloukalová M., Sýkora J., Císařová I., Cibulka R., Favereau L., Crassous J., Bilewicz R., Hrbáč J., Storch J., Žádný J., Vacek J.:
Flavin-Helicene Amphiphilic Hybrids: Synthesis, Characterization, and Preparation of Surface-Supported Films
ChemPlusChem 2021, 86, 982-990. DOI: 10.1002/cplu.202100092
- Tolba A. H., Krupička M., Chudoba J., Cibulka R.:
mide bond formation via aerobic photooxidative coupling of aldehydes with amines catalyzed by a riboflavin derivative
Org. Lett. 2021, 23, 6825-6830. DOI: 10.1021/acs.orglett.1c02391
- Tetour, D.; Novotná, M.; Hodačová, J.:
Enantioselective Henry Reaction Catalyzed by Copper(II) Complex of Bis(trans-cyclohexane-1,2-diamine)-Based Ligand.
Catalysts 2021, 11, 41. http:\\dx.doi.org/10.3390/catal11010041
- Cadart, T.; Nečas, D.; Kaiser, R. P.; Favereau, L.; Císařová, I.; Gyepes, R.; Hodačová, J.; Kalíková, K.; Bednárová, L.; Crassous, J.; Kotora, M.:
Rhodium-Catalyzed Enantioselective Synthesis of Highly Fluorescent and CPL Active Dispiroindeno[2,1‐c]fluorenes.
Chem. Eur. J. 2021, 27, 11279-11284.https://doi.org/10.1002/chem.202100759 - Tetour, D.; Paška, T.; Máková, V.; Nikendey Holubová, B.; Karpíšková, J.; Řezanka, M.; Brus, J.; Hodačová, J.:
Cinchonine-based organosilica materials as heterogeneous catalysts of enantioselective alkene dihydroxylation.
J. Catal. 2021, 404, 493-500. https://doi.org/10.1016/j.jcat.2021.10.015 - Kortus, D.; Krizova, K.; Dvorakova, H.; Eigner, V.; Lhotak, P.:
Synthesis of 2,8-dithiacalix[4]arene based on fragment condensation
Tetrahedron Lett. 2021, 69, 152924 DOI:10.1016/j.tetlet.2021.152924 - Kortus, D.; Eigner, V.; Lhotak, P.:
Regio- and stereoselectivity of spirodienone formation in 2,14-dithiacalix[4]arene
New J. Chem. 2021, 45, 8563-8571 DOI:10.1039/d1nj01257f - Kortus, D.; Kundrat, O.; Cejka, J.; Dvorakova, H.; Lhotak, P.:
Chemistry of 2,14-Dithiacalix[4]arene: Searching for the Missing Fifth Conformer
J. Org. Chem. 2021 DOI:10.1021/acs.joc.1c01173 - Curinova, P.; Winkler, M.; Krupkova, A.; Cisarova, I.; Budka, J.; Wun, C.N.; Blechta, V.; Maly, M.; Stastna-Cervenkova, L.; Sykora, J.; Strasak, T.:
Transport of Anions across the Dialytic Membrane Induced by Complexation toward Dendritic Receptors
ACS Omega 2021, 23, 15514-15522 DOI:10.1021/acsomega.1c02142 - Hartman T., Reisnerová M., Chudoba J., Svobodová E., Archipowa N., Kutta R. J., Cibulka R.:
Photocatalytic Oxidative [2+2] Cycloelimination Reactions with Flavinium Salts: Mechanistic Study and Influence of the Catalyst Structure
ChemPlusChem 2021, 86, 373–386. DOI: 10.1002/cplu.202000767
COVER:
- Edlová, T.; Čubiňák, M.; Tobrman, T.:
Cross-Coupling Reactions of Double or Triple Electrophilic Templates for Alkene Synthesis.
Synthesis 2021, 53, 255-266. DOI: 10.1055/s-0040-1707270 - Tobrman, T.:
Synthesis of 2-Substituted Cyclobutanones by a Suzuki Reaction and Dephosphorylation Sequence
Eur. J. Org. Chem. 2021, 3260-3269 DOI:10.1002/ejoc.202100464 - Tobrman, T.:
Substrate-Controlled Regioselective Bromination of 1,2-Disubstituted Cyclobutenes: An Application in the Synthesis of 2,3-Disubstituted Cyclobutenones
J. Org. Chem. 2021, 86, 5820-5831 DOI:10.1021/acs.joc.1c00261 - ; Tobrman, T.; J
- Skopalová H., Kozmík V., Šmahel M., Svoboda J., Pacherová O., Kohout M.:
Mesomorphic properties of non-symmetric bent-core liquid crystals with a lateral substituent in the apex position
Liq. Cryst. 2021, accepted. https://doi.org/10.1080/02678292.2020.1836567 - Cigl M., Hampl F., Svoboda J., Podoliak N., Stulov S., Kohout M., Novotná V.:
Laterally substituted biphenyl benzoates – synthesis and mesomorphic properties
Liq. Cryst. 2021, accepted. https://doi.org/10.1080/02678292.2020.1794069 - Spálovská D., Paškan M., Jurásek B., Kuchař M., Kohout M., Setnička V.:
Structural spectroscopic study of enantiomerically pure synthetic cathinones and their major metabolites
New J. Chem. 2021, 45, 850. https://doi.org/10.1039/D0NJ05065B - Wolrab D., Frühauf P., Kolderová N., Kohout M.:
Strong cation- and zwitterion-exchange-type mixed-mode stationary phases for separation of pharmaceuticals and biogenic amines in different chromatographic modes
J. Chromatogr. A 2021, 1635, 461751. https://doi.org/10.1016/j.chroma.2020.461751 - Linhart I., Hanzlíková I., Mráz J., Dušková Š., Tvrdíková M., Vachová H.:
Novel aminoarylcysteine adducts in globin of rats dosed with naphthylamine and nitronaphthalene isomers.
Arch. Toxicol. 2021, 95, 79-89. doi: 10.1007/s00204-020-02907-y - Marques M.M.; Beland F.A.; Lachenmeier D.W.; Phillips D.H.; Chung F.L.; Dorman D.C.; Elmore S.E.; Hammond S.K.; Krstev S.K.; Linhart I.; Long A.S.; Mandrioli D.; Ogawa K.; Pappas J.J.; Parra Morte J.M.; Talaska G.; Tang M.S.; Thakur N.; van Tongeren M.; Vineis P.:
IARC Monographs Vol 128 group. Carcinogenicity of acrolein, crotonaldehyde, and arecoline.
Lancet Oncol. 2021, 22, 19-20. doi: 10.1016/S1470-2045(20)30727-0
2020
- Poryvai A., Bubnov A., Kohout M.:
Chiral Photoresponsive Liquid Crystalline Materials Derived from Cyanoazobenzene Central Core: Effect of UV Light Illumination on Mesomorphic Behavior
Crystals 2020, 10, 1161. https://doi.org/10.3390/cryst10121161 - Skopalová H., Špaček P., Kozmík V., Svoboda J., Novotná V., Pociecha D., Kohout M.:
The Role of Substitution in the Apex Position of the Bent-Core on Mesomorphic Properties of New Series of Liquid Crystalline Materials
Crystals, 2020, 10, 735. https://doi.org/10.3390/cryst10090735 - Guselnikova O., Postnikov P., Kolská Z., Záruba K., Kohout M., Elashnikov R., Švorčík V., Lyutakov O.:
Homochiral metal-organic frameworks functionalized SERS substrate for atto-molar enantio-selective detection
Appl. Mater. Today 2020, 20, 100666. https://doi.org/10.1016/j.apmt.2020.100666 - Kolderová N., Jurásek B., Kuchař M., Lindner W., Kohout M.:
Gradient supercritical fluid chromatography coupled to mass spectrometry with a gradient flow of make-up solvent for enantioseparation of cathinones
J. Chromatogr. A 2020, 1625, 461286. https://doi.org/10.1016/j.chroma.2020.461286 - Šmahel M., Poryvai A., Xiang Y., Pociecha D., Troha T., Novotná V., Svoboda J., Kohout M.:
Photosensitive bent-core nematic liquid crystals with various linking units in the side arms: Structure-properties relationships
J. Mol. Liq. 2020, 306, 112743. https://doi.org/10.1016/j.molliq.2020.112743 - Cigl M., Jurok R., Hampl F., Svoboda J., Podoliak N., Novotná V.:
Lateral substituted phenyl biphenylcarboxylates ‒ non-chiral analogues of ferroelectric liquid crystals
Liq. Cryst. 2020, 47, 768-776. https://doi.org/10.1080/02678292.2019.1679903 - Gaálová J., Yalcinkaya F., Cuřínová P., Kohout M., Yalcinkaya B., Koštejn M., Jirsák J., Stibor I., Bara J. E., Van der Bruggen B., Izák P.:
Separation of racemic compound by nanofibrous composite membranes with chiral selector
J. Membr. Sci. 2020, 596, 117728. https://doi.org/10.1016/j.memsci.2019.117728 - Otmar M., Gaálová J., Žitka J., Brožová L., Cuřínová P., Kohout M., Hovorka Š., Bara J. E., Van der Bruggen B., Jirsák J., Izák P.:
Preparation of PSEBS membranes bearing (S)-(-)-methylbenzylamine as chiral selector
Eur. Polym. J. 2020, 122, 109381. https://doi.org/10.1016/j.eurpolymj.2019.109381 - Oeser, P.; Koudelka, J.; Dvořáková, H.; Tobrman, T.:
Formation of trisubstituted buta-1,3-dienes and α,β-unsaturated ketones via the reaction of functionalized vinyl phosphates and vinyl phosphordiamidates with organometallic reagents.
RSC Adv. 2020, 10, 35109-35120. DOI: 10.1039/D0RA07472A - Čubiňák, M.; Tobrman, T.:
Room-Temperature Negishi Reaction of Trisubstituted Vinyl Phosphates for the Synthesis of Tetrasubstituted Alkenes.
J. Org. Chem. 2020, 85, 10728-10739. DOI: 10.1021/acs.joc.0c01254 - Polák, P.; Čejka, J.; Tobrman, T.:
Formal Transition-Metal-Catalyzed Phosphole C–H Activation for the Synthesis of Pentasubstituted Phospholes.
Org. Lett. 2020, 22, 2187─2190 DOI: 10.1021/acs.orglett.0c00359 - Tobrman, T; Krupička, M.; Polák, P.; Dvořáková, H.; Čubiňák, M.; Babor, M.; Dvořák, D.:
Diastereoselective Cyclopropanation through Michael Addition‐Initiated Ring Closure between α,α‐Dibromoketones and α,β‐Unsaturated Fischer Carbene Complexes.
Eur. J. Org. Chem. 2020, 429─436 DOI: 10.1002/ejoc.201901503 - Guricová, M.; Tobrman, T.; Pižl, M.; Žižková, S.; Hoskovcová, I.; Dvořák, D.:
Synthesis, characterisation and electrochemical properties of Cr(0) aminocarbene complexes containing condensed heteroaromatic moiety.
Organomet. Chem. 2020, 905, 121023 DOI: 10.1016/j.jorganchem.2019.121023
- Kortus, D.; Kundrat, O.; Tlusty, M.; Cejka, J.; Dvorakova, H.; Lhotak, P.:
Inherent chirality through a simple dialkylation of 2,14-dithiacalix[4]arene
New J. Chem. 2020, 44, 14496-14504 DOI:10.1039/d0nj03468a - Tlusty, M.; Spalovska, D.; Kohout, M.; Eigner, V.; Lhotak, P.:
Ketone transformation as a pathway to inherently chiral rigidified calix[4]arenes
Chem. Commun. 2020, 56, 12773-12776 DOI:10.1039/d0cc05352j - Kolarikova, V.; Rybackova, M.; Svoboda, M.; Kvicala, J.:
Ring-closing metathesis of prochiral oxaenediynes to racemic 4-alkenyl-2-alkynyl-3,6-dihydro-2H-pyrans
Beilstein J. Org. Chem. 2020, 16, 2757–2768 DOI:10.3762/bjoc.16.226 - Simunek, O.; Rybackova, M.; Svoboda, M.; Kvicala, J.:
Synthesis, catalytic activity and medium fluorous recycle of fluorous analogues of PEPPSI catalysts
J. Fluorine Chem. 2020, 236, 109588 DOI:10.1016/j.jfluchem.2020.109588 - Graml, A.; Neveselý, T.; Kutta, R.-J.; Cibulka, R.; König, B.:
Deazaflavin reductive photocatalysis involves excited semiquinone radicals
Nature Comm. 2020, 11, 3174. DOI: 10.1038/s41467-020-16909-y - Regioselective SNAr reaction of the phenoxathiin-based thiacalixarene as a route to a novel macrocyclic skeleton
Chem. Commun. 2020, 56, 78-81 DOI:10.1039/c9cc08335a - Tlusty, M.; Dvorakova, H.; Cejka, J.; Kohout, M.; Lhotak, P.:
Regioselective formation of the quinazoline moiety on the upper rim of calix[4]arene as a route to inherently chiral systems
New J. Chem. 2020, 44, 6490-6500 DOI:10.1039/d0nj01035a - Máková, V.; Holubová, B.; Tetour, D.; Brus, J.; Řezanka, M.; Rysová, M.; Hodačová, J.:
(1S,2S)‐Cyclohexane‐1,2‐diamine‐based Organosilane Fibres as a Powerful Tool Against Pathogenic Bacteria
Polymers 2020, 12, 206 DOI:10.3390/polym12010206 - Cibulka, R.:
Strong chemical reducing agents produced by light
Nature 2020, 580, 31-32 DOI: 10.1038/d41586-020-00872-1 - Tolba, A. H.; Vávra, F.; Chudoba, J.; Cibulka, R.:
Tuning flavin-based photocatalytic systems for application in the mild chemoselective aerobic oxidation of benzylic substrates
Eur. J. Org. Chem. 2020, 1579–1585 DOI:10.1002/ejoc.201901628 Special issue Photochemical synthesis
- Schlosser J., Cibulka R., Groß P., Ihmels H., Mohrschladt C. J.:
Visible-light induced di-π-methane rearrangement of dibenzobarrelene derivatives
ChemPhotoChem 2020, 4, 132-137 DOI: 10.1002/cptc.201900221
- Zelenka, J.; Cibulka, R., Roithová, J.:
Flavinium catalyzed photooxidation: Detection and characterization of elusive peroxyflavinium intermediates
Angew. Chem. Int. Ed. 2020, 58, 15412-15420 DOI: 10.1002/anie.201906293
COVER:
- Mráz J., Hanzlíková I., Dušková Š., Tvrdíková M., Linhart I.:
N-(2-Hydroxyethyl)-l-valyl-l-leucine: a novel urinary biomarker of ethylene oxide exposure in humans.
Toxicol. Lett. 2020, 15, 18-22. doi: 10.1016/j.toxlet.2020.03.004
2019
- Slavik, P.; Krupicka, M.; Eigner, V.; Vrzal, L.; Dvorakova, H.; Lhotak, P.:
Rearrangement of meta-Bridged Calix[4]arenes Promoted by Internal Strain
J. Org. Chem. 2019, 84, 4229-4235, DOI:10.1021/acs.joc.9b00107 - Kortus, D.; Miksatko, J.; Kundrat, O.; Babor, M.; Eigner, V.; Dvorakova, H.; Lhotak, P.:
Chemistry of 2,14-Dithiacalix[4]arene: Alkylation and Conformational Behavior of Peralkylated Products
J. Org. Chem. 2019, 84, 11572-11580, DOI:10.1021/acs.joc.9b01493 - Tlusty, M.; Eigner, V.; Babor, M.; Kohout, M.; Lhotak, P.:
Synthesis of Upper Rim-double-bridged Calix[4]arenes bearing Seven Membered Rings and Related Compounds
RSC Advances 2019, 9, 22017-22030 DOI:10.1039/c9ra05075b - Pokluda, A.; Kohout, M.; Chudoba, J.; Krupička, M.; Cibulka, R.:
Nitrosobenzene – Reagent for the Mitsunobu Esterification Reaction
ACS Omega 2019, 4, 5012-5018. DOI: 10.1021/acsomega.8b03551 - Valenta, L.; Kovaricek, P.; Vales, V.; Bastl, Z.; Drogowska, K. A.; Verhagen, T. A.; Cibulka, R.; Kalbac, M.:
Spatially resolved covalent functionalization patterns on graphene
Angew. Chem. Int. Ed. 2019, 58, 1324-1328 DOI: 10.1002/anie.201810119 - Zelenka, J.; Svobodová, E.; Tarábek, J.; Hoskovcová, I.; Boguschová, V.; Bailly, S.; Sikorski, M.; Roithová, J.; Cibulka, R.:
Combining flavin photocatalysis and organocatalysis: metal-free aerobic oxidation of unactivated benzylic substrates
Org. Lett. 2019, 21, 114-119 DOI: 10.1021/acs.orglett.8b03547 - Jing, H.; Xu, M.; Xiang, Y.; Wang, E.; Liu, D.; Poryvai, A.; Kohout, M.; Éber, N.; Buka, A.:
Light tunable gratings based on flexoelectric effect in photoresponsive bent-core nematics.
Adv. Opt. Mater. 2019, 1801790 DOI: 10.1002/adom.201801790 - Pokluda, A.; Kohout, M.; Chudoba, J.; Krupička, M.; Cibulka, R.:
Nitrosobenzene: Reagent for the Mitsunobu esterification reaction.
ACS Omega 2019, 4, 5012-5018 DOI: 10.1021/acsomega.8b03551 - Spálovská, D.; Maříková, T.; Kohout, M.; Králík, F.; Kuchař, M.; Setnička, V.:
Methylone and pentylone: Structural analysis of new psychoactive substances.
Forensic Toxicol. 2019, 37, 366-377 DOI: 10.1007/s11419-019-00468-z - Poryvai, A.; Bubnov, A.; Pociecha, D.; Svoboda, J.; Kohout, M.:
The effect of the length of terminal n-alkyl carboxylate chain of self-assembling and photosensitive properties of chiral lactic acid derivatives.
J. Mol. Liq. 2019, 275, 829-838 DOI: 10.1016/j.molliq.2018.11.058 - Geibel, C.; Dittrich, K.; Woiwode, U.; Kohout, M.; Zhang, T.; Lindner, W.; Lämmerhofer, M.:
Evaluation of superficially porous particle based zwitterionic chiral ion exchangers against fully porous particle benchmarks for enantioselective ultra-high performance liquid chromatography.
J. Chromatogr. A 2019, 1603, 130-140 DOI: 10.1016/j.chroma.2019.06.026 - Poryvai, A.; Vojtylová-Jurkovičová, T.; Šmahel, M.; Kolderová, N.; Tomášková, P.; Sýkora, D.; Kohout, M.:
Determination of optical purity of lactic acid-based chiral liquid crystals and corresponding building blocks by chiral high-performance liquid chromatography and supercritical fluid chromatography.
Molecules 2019, 24, 1099 DOI: 10.3390/molecules24061099 - Tlustý, M.; Eigner, V.; Babor, M.; Kohout, M.; Lhoták, P.:
Synthesis of upper rim-double-bridged calix[4]arenes bearing seven membered rings and related compounds.
RSC Adv. 2019, 9, 22017-22030. DOI: 10.1039/C9RA05075B - Mráz J., Hanzlíková I., Linhart I., Dušková Š., Dabrowská L., Hejl K.:
N-(2-Hydroxyethyl)-L-valyl-L-leucine in rat urine as a hydrolytic cleavage product of ethylene oxide adduct with globin
Arch. Toxicol. 2019, 93, 603-613. doi: 10.1007/s00204-019-02388-8 - Linhart, I.:
Chemická legislativa pohledem laboratorního chemika.
Chemické listy 2019, 113, 140-148.
2018
- Zelenka, J.; Svobodová, E.; Tarábek, J.; Hoskovcová, I.; Boguschová, V.; Bailly, S.; Sikorski, M.; Roithová, J.; Cibulka, R.:
Combining flavin photocatalysis and organocatalysis: metal-free aerobic oxidation of unactivated benzylic substrates
Org. Lett. 2019, asap, DOI: 10.1021/acs.orglett.8b03547 - Jurásek, B.; Králík, F.; Rimpelová S.; Čejka, J.; Setnička, V.; Ruml, T.; Kuchař, M.; Kohout, M.:
Synthesis, Absolute Configuration and in vitro Cytotoxicity of Deschloroketamine Enantiomers: Rediscovered and Abused Dissociative Anaesthetic
New. J. Chem. 2018, 42, 19360-19368 DOI: 10.1039/c8nj03107j
COVER
- Landovsky, T.; Dvorakova, H.; Eigner, V.; Babor, M.; Krupicka, M.; Kohout, M.; Lhotak, P.:
Chemoselective Oxidation of Phenoxathiin-based Thiacalix[4]arene and the Stereoselective Alkylation of Products
New J. Chem. 2018, 42, 20074-20086 DOI: 10.1039/C8NJ04690E - Mojr, V.; Pitrova, G.; Strakova, K.; Prukala, D.; Brazevic, S.; Svobodová, E.; Hoskovcová, I.; Burdziński, G.; Slanina, T.; Sikorski, M.; Cibulka, R.:
Flavin Photocatalysts for visible Light [2+2] Cycloadditions: Structure, Reactivity and Reaction Mechanism
ChemCatChem 2018, 10, 849-858 DOI: 10.1002/cctc.201701490 - Marz, M.; Kohout, M.; Nevesely, T.; Chudoba, J.; Prukala, D.; Nizinski, S.; Sikorski, M.; Burdzinski, G.; Cibulka, R.:
Azodicarboxylate-free esterification with triphenylphosphine mediated by flavin and visible light: method development and stereoselectivity control
Org. Biomol. Chem. 2018, 16, 6809-6817 DOI:10.1039/C8OB01822G - Mundil, R.; Sokolohorskyj, A.; Hosek, J.; Cvacka, J.; Cisarova, I.; Kvicala, J.; Merna, J.:
Nickel and palladium complexes with fluorinated alkyl substituted α-diimine ligands for living/controlled olefin polymerization
Polym. Chem. 2018, 9, 1234-1248 DOI: 10.1039/C8PY00201K - Hosek, J.; Simunek, O.; Lipovska, P.; Kolarikova, V.; Kucnirova, K.; Edr, A.; Stepankova, N.; Rybackova, M.; Cvacka, J.; Kvicala, J.:
Medium Fluorous Separation Using Hydrofluoroether and Weakly Polar Solvents for Environmentally Friendly Recycling of Catalysts
ACS Sustainable Chem. Eng. 2018, 6, 7026-7034 DOI:10.1021/acssuschemeng.8b00865 - Kucnirova, K.; Simunek, O.; Rybackova, M.; Kvicala, J.:
Structural Assignment of Fluorocyclobutenes by 19F NMR Spectroscopy – Comparison of Calculated 19F NMR Shielding Constants with Experimental 19F NMR Shifts
Eur. J. Org. Chem. 2018, 3867-3874 DOI:10.1002/ejoc.201800482 - Slavik, P.; Eigner, V.; Lhotak, P.:
meta-Bridged calix[4]arenes with the methylene moiety possessing in/out stereochemistry of substituents
New J. Chem. 2018 asap DOI:10.1039/c8nj02577k - Landovsky, T.; Tichotova, M.; Vrzal, L.; Budka, J.; Eigner, V.; Dvorakova, H.; Lhotak, P.:
Structure elucidation of phenoxathiin-based thiacalix[4]arene conformations using NOE and RDC data
Tetrahedron 2018, 74, 902-907 DOI:10.1016/j.tet.2018.01.020 - Slavik, P.; Dvorakova, H.; Krupicka, M.; Lhotak, P.:
Unexpected cleavage of upper rim-bridged calix[4]arenes leading to linear oligophenolic derivatives
Org. Biomol. Chem. 2018, 59, 838-843 DOI:10.1039/C7OB03101G - Slavik, P.; Lhotak, P.:
Unusual reactivity of upper-rim bridged calix[4]arenes - Friedel-Crafts alkylation via cleavage of the macrocyclic skeleton
Tetrahedron 2018, 59, 1757-1759 DOI:10.1016/j.tetlet.2018.03.072 - Schmitt, K.; Woiwode, U.; Kohout, M.; Zhang, T.; Lindner, W.; Lämmerhofer, M.:
Comparison of small size fully porous particles and superficially porous particles of chiral anion-exchange type stationary phases in ultra-high performance liquid chromatography: effect of particle and pore size on chromatographic efficiency and kinetic performance
J. Chromatogr. A 2018, 1569, 149-159 DOI:10.1016/j.chroma.2018.07.056
- Spálovská, D.; Kralik, F.; Kohout, M.; Jurasek, B.; Habartová, L.; Kuchar, M.; Setnicka, V.:
Structure determination of butylone as a new psychoactive substance using chiroptical and vibrational spectroscopies
Chirality 2018, 30, 548-559 DOI:10.1002/chir.22825 - Wolrab, D.; Kohout, M.:
Multimodální stacionární fáze pro kapalinovou chromatografii, způsob jejich přípravy a jejich použití.
Národní patent č. 307339, číslo přihlášky 2017-193, datum udělení 2.5.2018, datum zveřejnění 13.6.2018. - Kohout, M.; Wernisch, S.; Tuma, J.; Hettegger, H.; Picha, J.; Lindner W.:
Effect of different immobilization strategies on chiral recognition properties of Cinchona-based anion exchangers
J. Sep. Sci. 2018, 41, 1355-1364 DOI:10.1002/jssc.201701213 - Bajtai, A.; Fekete, B.; Palkó, M.; Fülöp, F.; Lindner, W.; Kohout, M.; Ilisz, I.; Péter, A.:
Comparative study on the liquid chromatographic enantioseparation of cyclic β-amino acids and the related cyclic β-aminohydroxamic acids on Cinchona alkaloid-based zwitterionic chiral stationary phases
J. Sep. Sci. 2018, 41, 1216-1223 DOI:10.1002/jssc.201701190 - Sardella, R.; Macchiarulo, A.; Urbinati, F.; Ianni, F.; Carotti, A.; Kohout, M.; Lindner, W.; Péter, A.; Ilisz, I.:
Exploring the enantiorecognition mechanism of Cinchona alkaloid-based zwitterionic chiral stationary phases and the basic trans-paroxetine enantiomers
J. Sep. Sci. 2018, 41, 1199-1207 DOI:10.1002/jssc.201701068 - Tuma, J.; Kohout, M.:
Silica gel-immobilized multidisciplinary materials applicable in stereoselective organocatalysis and HPLC separation
RSC Adv. 2018, 8, 1174-1181 DOI:10.1039/c7ra12658a - Kozmik, V.; Rodinová, E.; Prausová, T.; Svoboda, J.; Novotná, V.; Pociecha, D.:
Mesogens with central naphthalene core substituted at various positions
Liq. Cryst. 2018, 45, 744-756 DOI: 10.1080/02678292.2017.1380238 - Bajzíková, K.; Vesely, J.; Kozmik, V.; Svoboda, J.; Novotná, V.; Pociecha, D.:
Thiophene central core for the design of bent-shaped liquid crystals
J. Mol. Liq. 2018, 267, 496-503 DOI:10.1016/j.molliq.2018.02.009 - Horcic, M.; Svoboda, J.; Novotná, V.; Pociecha, D.; Gorecka, E.:
Bent-core dimers with top-to-bottom linkage between central units
RSC Adv. 2018, 8, 22974-22985 DOI: 10.1039/c8ra04108c - Mráz J., Hanzlíková I., Dušková Š., Tvrdíková M., Chrástecká H., Vajtrová R., Linhart I.:
Determination of N-(2-hydroxyethyl)valine in globin of ethylene oxide-exposed workers using total acidic hydrolysis and HPLC-ESI-MS2
Toxicol. Lett. 2018, 298, 76-80. doi: 10.1016/j.toxlet.2018.06.1212
2017
- Maixner, J.; Jurásek, B.; Kohout, M.; Kuchar, M.; Kacer, P.:
X-ray powder diffraction data for (S)-Deschloroketamine hydrochloride, C13H18ClNO.
Powder Diffr. 2017, 32, 193-195. DOI:10.1017/S0885715617000586 - Bajzíková, K.; Svoboda, J.; Novotná, V.; Pociecha, D.; Gorecka E.:
Bent-core mesogens with an aromatic unit at the terminal position
New J. Chem. 2017, 41, 4672-4679 DOI: 10.1039/C6NJ03908A
- Tlusty, M.; Slavik, P.; Kohout, M.; Eigner, V.; Lhotak, P.:
Inherently Chiral Upper-Rim-Bridged Calix[4]arenes Possessing a Seven Membered Ring
Org. Lett. 2017, 19, 2933-2936 DOI:10.1021/acs.orglett.7b01170
- Jirasek, M.; Strakova, K.; Nevesely, T.; Svobodova, E.; Rottnerova, Z.; Cibulka, R.:
Flavin-mediated visible light [2+2] photocycloaddition of nitrogen and sulfur-containing dienes
Eur. J. Org. Chem. 2017, 2139-2146 DOI:10.1002/ejoc.201601377 - Slavik, P.; Eigner, V.; Bohm, S.; Lhotak, P.:
Mercuration of Calix[4]arene Immobilized in the 1,2- and 1,3-Alternate Conformations
Tetrahedron Letters 2017, 58, 1846-1850 DOI:10.1016/j.tetlet.2017.03.085 - Miksatko, J.; Eigner, V.; Kohout, M.; Lhotak, P.:
Regio-/stereoselective Formation of Monosulfoxides from Thiacalix[4]arenes in All Possible Conformations
Tetrahedron Letters 2017, 58, 1687-1691 DOI:10.1016/j.tetlet.2017.03.043 - März, M.; Chudoba, J.; Kohout, M.; Cibulka, R.:
Photocatalytic Esterification under Mitsunobu Reaction Conditions Mediated by Flavin and Visible Light
Org. Biomol. Chem. 2017, 15, 1970-1975 DOI:10.1039/C6OB02770A - Spackova, J.; Svobodova, E.; Hartman, T.; Stibor, I.; Kopecka, J.; Cibulkova, J.; Chudoba, J.; Cibulka, R.:
Visible light light [2+2] Photocycloaddition mediated by flavin derivative immobilized on mesoporous silica
ChemCatChem. 2017, 9, 1177-1181 DOI:10.1002/cctc.201601654 - Tlusty, M.; Slavik, P.; Dvorakova, H.; Eigner, V.; Lhotak, P.:
Synthesis and Study of Calix[4]arenes bearing Azo Moieties at the meta Position
Tetrahedron 2017, 73, 1230-1237 DOI:10.1016/j.tet.2017.01.025 - Rezankova, M.; Budka, J.; Miksatko, J.; Eigner, V.; Cisarova, I.; Curinova, P.; Lhotak, P.:
Anion Receptors based on Intramolecularly Bridged Calix[4]arenes bearing Ureido Functions
Tetrahedron 2017, 73, 742–749 DOI:10.1016/j.tet.2016.12.054
2016
- Slavik, P.; Eigner, V.; Lhotak, P.:
A General Method for Obtaining Calixarene Derivatives in the 1,2-Alternate Conformation
Tetrahedron 2016, 72, asap DOI:10.1016/j.tet.2016.08.028 - Klejch, T.; Slavicek, J.; Hudecek, O.; Eigner, V.; Gutierrez, N. A.; Curinova, P.; Lhotak, P.:
Calix[4]arene Containing a Ureido Functionality on the Lower Rim as Highly Efficient Receptors for Anion Recognition
New J. Chem. 2016, asap DOI:10.1039/c6nj01271j - Liska, A.; Lhotak, P.; Ludvik, J.:
Electrochemical Reduction and Intramolecular Electron Communication of Nitro Substituted Thiacalix[4]arene
Electroanalysis 2016, asap DOI:10.1002/elan.201600342 - Polak, P.; Vanova, H.; Dvorak, D.; Tobrman, T.:
Recent Progress in Transition Metal-Catalyzed Stereoselective Synthesis of Acyclic All-Carbon Tetrasubstituted Alkenes
Tetrahedron Lett. 2016, 57, 3684-3693 DOI:10.1016/j.tetlet.2016.07.030 - Navratil, R.; Tarabek, J.; Linhart, I.; Martinů, T.:
Radical and Nitrenoid Reactivity of 3-Halo-3-phenyldiazirines
Org. Lett. 2016, 18, 3734–3737 DOI:10.1021/acs.orglett.6b01753 - Miksatko, J.; Eigner, V.; Dvorakova, H.; Lhotak, P.:
Selective Oxidation of Thiacalix[4]arene (cone) to all Corresponding Sulfoxides
Tetrahedron Lett. 2016, 57, 3781–3784 DOI:10.1016/j.tetlet.2016.07.022 - Hartman, T.; Cibulka, R.:
Photocatalytic systems with flavinium salts: From photolyase models to synthetic tool for cyclobutane ring opening
Org. Lett. 2016, 18, 3710-3713 DOI:10.1021/acs.orglett.6b01743 - Bím, D.; Svobodová, E.; Eigner, V.; Rulíšek, L.; Hodačová, J.:
Copper(II) and Zinc(II) Complexes of Conformationally Constrained Polyazamacrocycles as Efficient Catalysts for RNA Model Substrate Cleavage in Aqueous Solution at Physiological pH
Chem. Eur. J. 2016, 22, 10426-10437 DOI:10.1002/chem.201601175
- Kohout, M.; Bubnov, A.; Šťurala, J.; Novotná, V.; Svoboda, J.:
Effect on alkyl chain length in the terminal ester group on mesomorphic properties of new chiral lactic acid derivatives
Liq. Cryst. 2016, 43, 1472-1485 DOI:10.1080/02678292.2016.1185170 - Grecsó, N.; Kohout, M.; Carotti, A.; Sardella, R.; Natalini, B.; Fülöp, F.; Lindner, W.; Péter, A.; Ilisz, I.:
Mechanistic considerations of enantiorecognition on novel Cinchona alkaloid-based zwitterionic chiral stationary phases from the aspect of the separation of trans-paroxetine enantiomers as model compounds
J. Pharm. Biomed. Anal. 2016, 124, 164-173 DOI:10.1016/j.jpba.2016.02.043 - Wolrab, D.; Frühauf, P.; Moulisova, A.; Kuchar, M.; Gerner, C.; Lindner, W.; Kohout, M.:
Chiral separation of new designer drugs (Cathinones) on chiral ion-exchange type stationary phases
J. Pharm. Biomed. Anal. 2016, 120, 306-315 DOI:10.1016/j.jpba.2015.12.023
- Slavik, P.; Eigner, V.; Lhotak, P.:
Shaping of Calix[4]arenes via Double Bridging of the Upper Rim
Cryst. Eng. Comm. 2016, 18, 4964-4970 DOI:10.1039/C6CE00314A
- Neveselý, T.; Svobodová, E.; Chudoba, J.; Sikorski, M.; Cibulka, R.:
Efficient metal-free aerobic photooxidation of sulfides to sulfoxides mediated by a vitamin B2 derivative and visible light
Adv. Synth. Catal. 2016, 358, 1654-1663 DOI:10.1002/adsc.201501123 - Mraz, J.; Hanzlikova, I.; Moulisova, A.; Duskova, S.; Hejl, K.; Bednarova, A.; Dabrowska, L.; Linhart, I.:
Hydrolytic cleavage products of globin adducts in urine as possible biomarkers of cumulative dose: Proof of concept using styrene oxide as a model adduct-forming compound
Chem. Res. Toxicol. 2016, 29, 676-686 DOI:10.1021/acs.chemrestox.5b00518 - Dolenský, B.; Kvíčala, J.; Paleta, O.:
Methyl fluoroalkanoate as methyl-transferring reagent. Unexpected participation of BAl2 (SN2) mechanism in the reaction of methyl 2,3,3,3-tetrafluoro-2-methoxypropanoate with amines.
J. Fluorine Chem. 2016, 185, 31-35. DOI:10.1016/j.jfluchem.2016.02.012 - Kundrat, O.; Slavik, P.; Miksatko, J.:
Introducing an Amine Group to Calix[5]arene: Comparison of Several Methods
Supramol. Chem. 2016, 28, 450-454, DOI:10.1080/10610278.2015.1119275 - Tůma, J.; Kohout, M.; Svoboda, J.; Novotná, V.; Pociecha, D.:
Bent-shaped liquid crystals based on 4-substituted 3-hydroxybenzoic acid central core - Part II
Liq. Cryst. 2016, 43, 547-563, DOI: 10.1080/02678292.2015.1125535 - Kotek, V.; Polák, P.; Tobrman, T.:
Efficient and Simple Preparation of Functionalized 1,1-Dibromoenol Phosphates
Monatshefte fuer Chemie 2016, 147, 405-412, DOI: 10.1007/s00706-015-1613-6 - Linhart, I.; Himl, M.; Zidkova, M.; Balikova, M.; Lhotakova, E.; Palenicek, T.:
Metabolic Profile of Mephedrone: Identification of Normephedrone Conjugates with Dicarboxylic Acids as a New Type of Xenobiotic Phase II Metabolites
Toxicology Letters 2016, 240, 114-121, DOI: 10.1016/j.toxlet.2015.10.025 - Botha, F.; Eigner, V.; Dvorakova, H.; Lhotak, P.:
Arylation of Thiacalix[4]arenes using Organomercurial Intermediates
New J. Chem. 2016, 40, 1104-1110, DOI:10.1039/C5NJ02427G + cover picture - Slavik, P.; Kohout, M.; Böhm, S.; Eigner, V.; Lhotak, P.:
Synthesis of Inherently Chiral Calixarenes via Direct Mercuration of the Partial Cone Conformation
Chem. Commun. 2016, 52, 2366-2369 DOI:10.1039/C5CC09388K - Stejskal, F.; Curinova, P.; Lhotak, P.:
Unexpected Formation of Disulfide-based Biscalix[4]arenes
Tetrahedron 2016, 72, 760-766 DOI:10.1016/j.tet.2015.12.037
2015
- Kotek, V.; Dvořáková, H.; Tobrman, T.:
Modular and Highly Stereoselective Approach to All-Carbon Tetrasubstituted Alkenes
Org. Lett. 2015, 17, 608-611. DOI: 10.1021/ol503624v - Hartman, T.; Šturala, J.; Cibulka, R.:
Two-phase Oxidations with Aqueous Hydrogen Peroxide Catalysed by Amphiphilic Pyridinium and Diazinium Salts
Adv. Synth. Catal. 2015, 357, 3573-3586. DOI:10.1002/adsc.201500687 - Kolaříková, V.; Šimůnek, O.; Rybáčková, M.; Cvačka, J.; Březinová, A.; Kvíčala, J.:
Transition Metal Complexes Bearing NHC Ligands with Secondary Polyfluoroalkyl Groups
J. Chem. Soc., Dalton Trans. 2015, 44, 19663-19673. DOI:10.1039/C5DT02258D - Králík, A.; Linhart, I.; Váňa, L.; Moulisová, A.:
Identification of New DNA Adducts of Phenylnitrenium
Chem. Res. Toxic. 2015, 28, 1317-1325. DOI:10.1021/acs.chemrestox.5b00120 - Kozmík, V.; Poznik, M.; Svoboda, J.; Frere, P.:
Dithieno[3,2-b:2 ',3 '-d]furan as a New Building Block for Fused Conjugated Systems
Tetrahedron Lett. 2015, 56, 6251-6253. DOI:10.1016/j.tetlet.2015.09.107 - Nunes, S.; Bürglová, K.; Hodačová, J.; Ferreira, R.; Carlos, L.; Almeida, P.; Cattoën, X.; Man, M.; Bermudez, V.:
Nanostructuring of Bridged Organosilane Precursors with Pendant Alkyl Chains
Eur. J. Org. Chem. 2015, 1218-1225. DOI:10.1002/ejic.201402673 - Zajicova, M.; Eigner, V.; Budka, J.; Lhotak, P.:
Intramolecular Bridging of Calix[4]arene Dialdoximes
Tetrahedron Lett. 2015, 56, 5529–5532 DOI:10.1016/j.tetlet.2015.08.032
- Vrzal, L.; Kratochvilova-Simanova, M.; Landovsky, T.; Polivkova, K.; Budka, J.; Dvorakova, H.; Lhotak, P.:
Application of RDC Enhanced NMR Spectroscopy in Structural Analysis of Thiacalix[4]arene Derivatives
Org. Biomol. Chem. 2015, 13, 9610-9618 DOI:10.1039/C5OB01424G - Stejskal, F.; Eigner, V.; Dvorakova, H.; Curinova, P.; Lhotak, P.:
Direct C-H Azidation of Calix[4]arene as a Novel Method to Access meta Substituted Derivatives
Tetrahedron Lett. 2015, 56, 5357-5361 DOI:10.1016/j.tetlet.2015.08.002 - Slavik, P.; Eigner, V.; Lhotak, P.:
Intramolecularly Bridged Calix[4]arenes with Pronounced Complexation Ability toward Neutral Compounds
Org. Lett. 2015, 17, 2788-2791 DOI:10.1021/acs.orglett.5b01200 - Liska, A.; Flidrova, K.; Lhotak, P.; Ludvik, J.:
Influence of Structure on Electrochemical Reduction of Isomeric Mono- and Di-, Nitro- or Nitrosocalix[4]arenes
Monatshefte fuer Chemie 2015, 146, 857-862 DOI:10.1007/s00706-015-1441-8 - Hucko, M.; Dvorakova, H.; Eigner, V.; Lhotak, P.:
2,14-Dithiacalix[4]arene and its Homooxa Analogues: Synthesis and Dynamic NMR Study of Conformational Behaviour
Chem. Comm. 2015, 51, 7051-7053 DOI:10.1039/C5CC00819K - Spendlikova, I.; John, J.; Cuba, V.; Jirasek, J.; Lhotak, P.:
Thiacalixarenes: Radiation Stability and Eu/Am Extraction in Synergistic Systems with COSANs
J. Radioanalyt. Nucl. Chem. 2015, 304, 257-262 DOI:10.1007/s10967-014-3694-9 - Mackova, M.; Miksatko, J.; Budka, J.; Eigner, V.; Curinova, P.; Lhotak, P.:
Chiral Anion Recognition by a Ureido-Thiacalix[4]arene Ligand Immobilized in the 1,3-Alternate Conformation
New J. Chem. 2015, 39, 1382-1389 DOI:10.1039/C4NJ01956C - Flidrova, K.; Liska, A.; Ludvik, J.; Eigner, V.; Lhotak, P.:
Fullerene Recognition by 5-Nitro-11,17,23,29-tetramethylcalix[5]arene
Tetrahedron Lett. 2015, 56, 1535-1538 DOI:10.1016/j.tetlet.2015.02.016 - Skacel, J.; Budka, J.; Eigner, V.; Lhotak, P.:
Regioselective Friedel-Crafts Acylation of Calix[4]arenes
Tetrahedron 2015, 71, 1959-1965 DOI:10.1016/j.tet.2015.02.021 - Trišović, N.; Antanasijević, J.; Tóth-Katona, T.; Kohout, M.; Salamonczyk, M.; Sprunt, S.; Jákli, A.; Fodor-Csorba, K.:
Azo-containing asymmetric bent-core liquid crystals with modulated smectic phase
RSC Adv. 2015, 5, 64886-64891. DOI: 10.1039/c5ra09764a
- Kohout, M.; Bielec, B.; Steindl, P.; Trettenhahn, G.; Lindner, W.:
Mechanistic aspects of the direct C-acylation of cyclic 1,3-diones with various unactivated carboxylic acids
Tetrahedron 2015, 71, 2698-2707. DOI: 10.1016/j.tet.2015.03.037
- von Koschitzky, I.; Gerhardt, H.; Lämmerhofer, M.; Kohout, M.; Gehringer, M.; Laufer, S.; Pink, M.; Schmitz-Spanke, S.; Strube, C.; Kaiser, A.:
New insights into novel inhibitors against deoxyhypusine hydrolase from plasmodium falciparum: compounds with an iron chelating potential
Amino Acids 2015, 47, 1155-1166. DOI: 10.1007/s00726-015-1943-z
- Kohout, M.; Kozmík, V.; Slabochová, M.; Tůma, J.; Svoboda, J.; Novotná, V.; Pociecha, D.:
Bent-shaped liquid crystals based on 4-substituted 3-hydroxybenzoic acid central core
Liq. Cryst. 2015, 42, 87-103. DOI: 10.1080/02678292.2014.965232
- Moulisová, A.; Linhart, I.:
Preparation of cysteine adducts by regioselective ring-opening reactions of phenyloxirane
Heterocyclic Commun. 2015, 21, 61-65 DOI:10.1515/hc-2015-0042 - Rybáčková, M.; Hošek, J.; Šimůnek, O.; Kolaříková, V.; Kvíčala, J.:
Computational study of productive and non-productive cycles in fluoroalkene metathesis.
Beilstein J. Org. Chem. 2015, 11, 2150–2157. DOI:10.3762/bjoc.11.232 - Hošek, J.; Rybáčková, M.; Čejka, J.; Cvačka, J.; Kvíčala, J.:
Synthesis of heavy Fluorous Ruthenium Metathesis catalysts using the stereoselective addition of polyfluoroalkyllithium to sterically hindered diimines.
Organometallics 2015, 34(13), 3327-3334. DOI:10.1021/acs.organomet.5b00325
- Křováček M., Dvořák D.:
Synthesis of potentially biologically active 6-(1,3-butadiynyl)purines.
J. Heterocyclic Chem. 2015, 52, 40-47 DOI:10.1002/jhet.1938 - Bousa, D.; Jankovsky, O.; Sedmidubsky, D.; Luxa, J.; Sturala, J.; Pumera, M.; Sofer, Z.:
Mesomeric Effects of Graphene Modified with Diazonium Salts: Substituent Type and Position Influence its Properties
Chem. Eur. J. 2015, 21, 17728-17738. DOI:10.1002/chem.201502127 - Tomanová P., Šturala J., Buděšínský M., Cibulka R.:
A click chemistry approach towards flavin-cyclodextrin conjugates – bioinspired sulfoxidation catalysts,
Molecules 2015, 20, 19837-19848. DOI:10.3390/molecules201119667 - Holakovský R., März M., Cibulka R.:
Urea derivatives based on a 1,1'-binaphthalene skeleton as chiral solvating agents for sulfoxides,
Tetrahedron: Asymmetry 2015, 26, 1328-1334. DOI:10.1016/j.tetasy.2015.10.011 - Mojr V., Svobodová E., Straková K., Neveselý T., Chudoba J., Dvořáková H., Cibulka R.:
Tailoring Flavins for Visible Light Photocatalysis: Organocatalytic [2+2] Cycloadditions Mediated by a Flavin Derivative and Visible Light,
Chem. Comm. 2015, 51, 12036 – 12039. DOI:10.1039/C5CC01344E - Sofer, Z.; Jankovský, O.; Šimek, P.; Sedmidubský, D.; Šturala, J.; Kosina, J.; Mikšová, R.; Macková, A.; Mikulics, M.; Pumera, M.:
Insight into the Mechanism of the Thermal Reduction of Graphite Oxide: Deuterium-Labeled Graphite Oxide Is the Key,
ACS Nano 2015, 9, 5478-5485. DOI:10.1021/acsnano.5b01463 - Šturala J., Boháčová S., Chudoba J., Metelková R., Cibulka R.:
Electron-deficient Heteroarenium salts – an Organocatalytic Tool for Activation of Hydrogen Peroxide in Oxidations,
J. Org. Chem. 2015, 80, 2676-2699. DOI:10.1021/jo502865f - Cibulka, R.:
Artificial flavin systems for chemoselective and stereoselective oxidations (Microreview),
Eur. J. Org. Chem. 2015, 915-932. DOI:10.1002/ejoc.201403275 + Cover picture