stdClass Object
(
[nazev] => Department of Organic Chemistry
[adresa_url] =>
[api_hash] =>
[seo_desc] =>
[jazyk] =>
[jednojazycny] =>
[barva] =>
[indexace] => 1
[obrazek] =>
[ga_force] =>
[cookie_force] =>
[secureredirect] =>
[google_verification] => UOa3DCAUaJJ2C3MuUhI9eR1T9ZNzenZfHPQN4wupOE8
[ga_account] => UA-10822215-3
[ga_domain] =>
[ga4_account] => G-VKDBFLKL51
[gtm_id] =>
[gt_code] =>
[kontrola_pred] =>
[omezeni] => 0
[pozadi1] =>
[pozadi2] =>
[pozadi3] =>
[pozadi4] =>
[pozadi5] =>
[robots] =>
[htmlheaders] =>
[newurl_domain] => 'uoch.vscht.cz'
[newurl_jazyk] => 'en'
[newurl_akce] => '[en]'
[newurl_iduzel] =>
[newurl_path] => 8548/6214/6521
[newurl_path_link] => Odkaz na newurlCMS
[iduzel] => 6521
[platne_od] => 31.10.2023 17:15:00
[zmeneno_cas] => 31.10.2023 17:15:57.546654
[zmeneno_uzivatel_jmeno] => Jan Kříž
[canonical_url] =>
[idvazba] => 7334
[cms_time] => 1714156178
[skupina_www] => Array
(
)
[slovnik] => stdClass Object
(
[logo_href] => /
[logo] =>  [google_search] => 001523547858480163194:u-cbn29rzve
[social_fb_odkaz] => https://www.facebook.com/uoch.vscht
[social_tw_odkaz] => https://twitter.com/uoch_vscht
[social_yt_odkaz] =>
[paticka_budova_a_nadpis] => BUILDING A
[paticka_budova_a_popis] => Rector,
Department of Communications,
Department of Education,
FCT Dean’s Office,
Centre for Information Services
[paticka_budova_b_nadpis] => BUILDING B
[paticka_budova_b_popis] => Department of R&D, Dean’s Offices:
FET,
FFBT,
FCE,
Computer Centre,
Department of International Relations,
Bursar
[paticka_budova_c_nadpis] => BUILDING C
[paticka_budova_c_popis] =>
Crèche Zkumavka,
General Practitioner,
Department of Economics and Management,
Department of Mathematics
[paticka_budova_1_nadpis] => NATIONAL LIBRARY OF TECHNOLOGY
[paticka_budova_1_popis] =>
[paticka_budova_2_nadpis] => CAFÉ CARBON
[paticka_budova_2_popis] =>
[paticka_adresa] => UCT Prague
[google_search] => 001523547858480163194:u-cbn29rzve
[social_fb_odkaz] => https://www.facebook.com/uoch.vscht
[social_tw_odkaz] => https://twitter.com/uoch_vscht
[social_yt_odkaz] =>
[paticka_budova_a_nadpis] => BUILDING A
[paticka_budova_a_popis] => Rector,
Department of Communications,
Department of Education,
FCT Dean’s Office,
Centre for Information Services
[paticka_budova_b_nadpis] => BUILDING B
[paticka_budova_b_popis] => Department of R&D, Dean’s Offices:
FET,
FFBT,
FCE,
Computer Centre,
Department of International Relations,
Bursar
[paticka_budova_c_nadpis] => BUILDING C
[paticka_budova_c_popis] =>
Crèche Zkumavka,
General Practitioner,
Department of Economics and Management,
Department of Mathematics
[paticka_budova_1_nadpis] => NATIONAL LIBRARY OF TECHNOLOGY
[paticka_budova_1_popis] =>
[paticka_budova_2_nadpis] => CAFÉ CARBON
[paticka_budova_2_popis] =>
[paticka_adresa] => UCT Prague
Technická 5
166 28 Prague 6 – Dejvice
IČO: 60461373 / VAT: CZ60461373
Czech Post certified digital mail code: sp4j9ch
Copyright: UCT Prague 2015
Information provided by the Department of Organic Chemistry. Technical support by the Computing Centre.
[paticka_odkaz_mail] => mailto:kundrato@vscht.cz
[social_fb_title] => Facebook of Department
[social_tw_title] => Twitter of Department
[social_yt_title] =>
[aktualizovano] => Updated
[autor] => Author
[paticka_mapa_alt] =>
[drobecky] => You are here: VŠCHT Praha – FCHT – ÚOCH
[intranet_odkaz] => http://intranet.vscht.cz/
[intranet_text] => Intranet
[logo_mobile_href] => /
[logo_mobile] =>  [mobile_over_nadpis_menu] => Menu
[mobile_over_nadpis_search] => Search
[mobile_over_nadpis_jazyky] => Languages
[mobile_over_nadpis_login] => Login
[menu_home] => Homepage
[zobraz_desktop_verzi] => switch to desktop version
[zobraz_mobilni_verzi] => switch to mobile version
[more_info] => more information
[paticka_mapa_odkaz] =>
[nepodporovany_prohlizec] => For full access, please use different browser.
[preloader] => Wait a second...
[social_in_odkaz] =>
[den_kratky_4] =>
[archiv_novinek] =>
[novinky_servis_archiv_rok] =>
[novinky_kategorie_1] =>
[novinky_kategorie_2] =>
[novinky_kategorie_3] =>
[novinky_kategorie_4] =>
[novinky_kategorie_5] =>
[novinky_archiv_url] =>
[novinky_servis_nadpis] =>
[novinky_dalsi] =>
[hledani_nadpis] => hledání
[hledani_nenalezeno] => Nenalezeno...
[hledani_vyhledat_google] => vyhledat pomocí Google
[social_li_odkaz] =>
)
[poduzel] => stdClass Object
(
[6525] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[6532] => stdClass Object
(
[obsah] =>
[iduzel] => 6532
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6530] => stdClass Object
(
[obsah] =>
[iduzel] => 6530
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6531] => stdClass Object
(
[obsah] =>
[iduzel] => 6531
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
)
[iduzel] => 6525
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6526] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[21923] => stdClass Object
(
[nazev] => Department of Organic Chemistry
[seo_title] => Department of Organic Chemistry
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] =>
[mobile_over_nadpis_menu] => Menu
[mobile_over_nadpis_search] => Search
[mobile_over_nadpis_jazyky] => Languages
[mobile_over_nadpis_login] => Login
[menu_home] => Homepage
[zobraz_desktop_verzi] => switch to desktop version
[zobraz_mobilni_verzi] => switch to mobile version
[more_info] => more information
[paticka_mapa_odkaz] =>
[nepodporovany_prohlizec] => For full access, please use different browser.
[preloader] => Wait a second...
[social_in_odkaz] =>
[den_kratky_4] =>
[archiv_novinek] =>
[novinky_servis_archiv_rok] =>
[novinky_kategorie_1] =>
[novinky_kategorie_2] =>
[novinky_kategorie_3] =>
[novinky_kategorie_4] =>
[novinky_kategorie_5] =>
[novinky_archiv_url] =>
[novinky_servis_nadpis] =>
[novinky_dalsi] =>
[hledani_nadpis] => hledání
[hledani_nenalezeno] => Nenalezeno...
[hledani_vyhledat_google] => vyhledat pomocí Google
[social_li_odkaz] =>
)
[poduzel] => stdClass Object
(
[6525] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[6532] => stdClass Object
(
[obsah] =>
[iduzel] => 6532
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6530] => stdClass Object
(
[obsah] =>
[iduzel] => 6530
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6531] => stdClass Object
(
[obsah] =>
[iduzel] => 6531
[canonical_url] => //uoch.vscht.cz
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
)
[iduzel] => 6525
[canonical_url] =>
[skupina_www] => Array
(
)
[url] =>
[sablona] => stdClass Object
(
[class] =>
[html] =>
[css] =>
[js] =>
[autonomni] =>
)
)
[6526] => stdClass Object
(
[obsah] =>
[poduzel] => stdClass Object
(
[21923] => stdClass Object
(
[nazev] => Department of Organic Chemistry
[seo_title] => Department of Organic Chemistry
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] => New PhD topics of our department: here
All publications of our authors (since 2015): here
[urlnadstranka] =>
[ogobrazek] =>
[pozadi] =>
[iduzel] => 21923
[canonical_url] =>
[skupina_www] => Array
(
)
[url] => /home
[sablona] => stdClass Object
(
[class] => boxy
[html] =>
[css] =>
[js] => $(function() {
setInterval(function () { $('*[data-countdown]').each(function() { CountDownIt('#'+$(this).attr("id")); }); },1000);
setInterval(function () { $('.homebox_slider:not(.stop)').each(function () { slide($(this),true); }); },5000);
});
function CountDownIt(selector) {
var el=$(selector);foo = new Date;
var unixtime = el.attr('data-countdown')*1-parseInt(foo.getTime() / 1000); if(unixtime<0) unixtime=0;
var dnu = 1*parseInt(unixtime / (3600*24)); unixtime=unixtime-(dnu*(3600*24));
var hodin = 1*parseInt(unixtime / (3600)); unixtime=unixtime-(hodin*(3600));
var minut = 1*parseInt(unixtime / (60)); unixtime=unixtime-(minut*(60));
if(unixtime<10) {unixtime='0'+unixtime;}
if(dnu<10) {unixtime='0'+dnu;}
if(hodin<10) {unixtime='0'+hodin;}
if(minut<10) {unixtime='0'+minut;}
el.html(dnu+':'+hodin+':'+minut+':'+unixtime);
}
function slide(el,vlevo) {
if(el.length<1) return false; var leva=el.find('.content').position().left; var sirka=el.width(); var pocet=el.find('.content .homebox').length-1;
var cislo=leva/sirka*-1; if(vlevo) { if(cislo+1>pocet) cislo=0; else cislo++; } else { if(cislo==0) cislo=pocet-1; else cislo--; }
el.find('.content').animate({'left':-1*cislo*sirka});
el.find('.slider_puntiky a').removeClass('selected');
el.find('.slider_puntiky a.puntik'+cislo).addClass('selected');
return false;
}
function slideTo(el,cislo) {
if(el.length<1) return false; var sirka=el.width(); var pocet=el.find('.content .homebox').length-1; if(cislo<0 || cislo>pocet) return false;
el.find('.content').animate({'left':-1*cislo*sirka});
el.find('.slider_puntiky a').removeClass('selected');
el.find('.slider_puntiky a.puntik'+cislo).addClass('selected');
return false;
}
[autonomni] => 1
)
)
[16022] => stdClass Object
(
[nazev] => Department of Organic Chemistry - Staff
[seo_title] => Department of Organic Chemistry
[seo_desc] =>
[autor] =>
[autor_email] =>
[perex] =>
[ikona] => telefon-zvoni
[obrazek] =>
[ogobrazek] =>
[pozadi] =>
[obsah] =>
| k Phonebook of the Department | |||
Professors: |
|||
|
Prof. Radek Cibulka e +420 22044 4182 d A 278c |
Head of Department | www | ResearcherID |
|
Prof. Pavel Lhoták e +420 22044 5055 d A 249 |
www | ||
|
Prof. Jaroslav Kvíčala e +420 22044 4240 d A 278d |
www | ||
|
Prof. Jiří Svoboda e +420 22044 3688 d A 278e |
www | ||
Associate Professors: |
|||
|
Assoc. Prof. Jan Budka e +420 22044 4284 d A 251 |
DOC web editor | www | |
|
Assoc. Prof. Jana Hodačová e +420 22044 4173 d A 270 |
www | ||
|
Assoc. Prof. Michal Kohout e +420 22044 5012 d A 258 |
www | ||
|
Assoc. Prof. Igor Linhart e +420 22044 4165 d A 266 |
www | ||
|
Assoc. Prof. Tomáš Tobrman e +420 22044 4245 d A 271 |
RIT | www | |
Assistant Professors: |
|||
|
Dr. Michal Himl e +420 22044 4165 d A 266 |
www | ||
|
Dr. Roman Holakovský e +420 22044 4279 d A 255 |
www | ||
|
Dr. Petr Kovaříček e +420 22044 2040 d B3 207 |
www | ||
|
Dr. Václav Kozmík e +420 22044 4118 d A 308 |
www | ||
|
Dr. Martin Krupička e +420 220 444 173 |
www | ||
|
Dr. Ondřej Kundrát e +420 22044 4280 d A 253 |
www | ResearcherID | |
|
Dr. Petra Ménová e +420 22044 3686 d A 268 |
www | ||
|
Dr. Pavla Perlíková e +420 22044 2039 d B3 206 |
www | ||
|
Dr. Markéta Rybáčková e +420 22044 4242 d A 259 |
Secretary of Department |
www | |
|
Dr. Eva Svobodová e +420 22044 4249 d A 260 |
www | ||
Technicians: |
|||
|
Květa Bártová e +420 22044 3686 d A 268 |
|||
|
Ivana Bocková, MSc. e +420 22044 4280 d A 250 |
|||
| Michaela Kadlecová
e +420 22044 4276 d A 262 b Michaela.Kadlecova@vscht.cz |
|||
|
Martina Kovandová e +420 22044 4279 d A 255 |
|||
|
Vladimír Kuneš e +420 22044 4277 d A L06 |
|||
|
Jana Netušilová e +420 22044 4164 d A 278a |
Administrative Support of Department | ||
|
Markéta Slabochová, MSc. e +420 22044 4059 d A 308 |
Economist of Department | ||
|
Helena Štenglová e +420 22044 4278 d A 257 |
|||
|
Ing. David Tetour e +420 22044 4173 d A270 |
|||
|
Marta Tokárová e +420 220 444 245 d A 271 |
|||
Essential Information for Current Students
Bachelor Students
Master Students
PhD Students
Lectures delivered by members of the Department
- Toxicology and ecology
- Organic chemistry I, II and III
- Structural analysis
- Reactivity of organic compounds
- Molecular design
- Organic synthesis I and II
- Quantum organic chemistry
- Farmacochemistry
- Organic stereochemistry
- Organometallic chemistry
- Organic chemistry of selected elements
DATA
stdClass Object
(
[nazev] =>
[seo_title] => Kohout Research Group
[seo_desc] =>
[autor] =>
[autor_email] =>
[obsah] => Smart organic materials group

The web page is currently under construction. Please check for updates.
Contacts
|
Prof. Jiří Svoboda building A, room 278e + 420 220 44 3688 |
Dr. Václav Kozmík building A, room 308b + 420 220 44 4118 |
|
Assoc. Prof. Michal Kohout building A, rooms 278e/308b + 420 220 44 3688/4118 |
|
People
Employees:
- Assoc. Prof. Michal Kohout
- Prof. Jiří Svoboda
- Dr. Václav Kozmík
- Markéta Slabochová, MSc.
Ph.D. students:
- Anna Poryvai, MSc.
- Jana Herciková, MSc.
- Michal Šmahel, MSc.
- Tereza Černá, MSc.
- Natalie Kolderová, MSc.
- Martin Paškan , MSc.
Students:
- Barbora Jansová
- Žaneta Javorská
- Pavel Mata
- Jakub Kňava
- Diana Jágerová
- Anežka Kuncová
- Magdalena Labíková
- Jakub Dávid Malina
- Anna Rejzková
- Petr Bečvář
- Silvie Fabiánová
- Marie Kloubcová
Research topics
Photosensitive liquid crystals
Biologically active substances
Continuous flow organocatalysis
Current Special Issues guest-edited by group members
Photosensitive Liquid Crystals
https://www.mdpi.com/journal/crystals/special_issues/photosensitive_liquid_crystals
Chiral Separation by Liquid Chromatography
https://www.mdpi.com/journal/separations/special_issues/chiral_chromatography
Photosensitive liquid crystals
Light Tunable Gratings Based on Flexoelectric Effect in Photoresponsive Bent‐Core Nematics
A new photoresponsive bent‐core nematic (BCN) material, which exhibits flexoelectric domains (FDs) driven by electric field, is reported. Unexpectedly, it is found that the morphologies of FDs can be controlled by irradiation with light fields. This light tunability is ascribed to the photoisomerization effect of the azo moiety within the BCN molecules, where the ratio of trans and cis isomers changes according to the parameters of the light field, resulting in adjustable electric threshold and periodicity of FDs. Based on this principle, a prototype of controllable optical grating is assembled, whose operation can be manipulated by the wavelength or intensity of light. Due to the easy, instant, and remote operation by light, this optical, contactless tunability has a great advantage over traditional electric control in tunable photonic devices.

Jing H. et al. Adv. Opt. Mater. 2019, 7, 1801790. https://doi.org/10.1002/adom.201801790
Magnetic liquid crystals
All-organic liquid crystalline radicals with a spin unit in the outer position of a bent-core system
All-organic paramagnetic liquid crystals offer the advantage of a long-range order of liquid crystalline phases and the magnetic properties of the individual molecules. In such systems, the magnetic properties can be modified by phase transition or the application of external fields. This manuscript reports on paramagnetic all-organic bent-core liquid crystals having the radical-bearing unit (TEMPO) in the terminal position of an elongating side arm. The mesomorphic properties of the materials are ensured by the optimized molecular structure. The paramagnetic nature of the mesogenic materials is investigated by electron paramagnetic resonance, the magnetic properties of the bulk materials are studied by SQUID magnetometry. It is shown that the materials preserve their magnetic properties within the whole temperature range of liquid crystalline behaviour. Moreover, a strong correlation between spin orientation and molecular alignment within different mesophases has been observed, however, SQUID measurements do not provide evidence about spin glass formation. Unlike materials presented thus far, the position of the spin unit has a plausible effect on the formation of mesophases leading to unique polymorphism of the studied paramagnetic compounds. For two six-ring hockey-stick-like compounds, polymorphism with four different mesophases, including two B1Rev-type phases, has been found in a broad temperature range (about 20 °C each). Thus, for the first time, such behaviour is described for all-organic paramagnetic bent-core liquid crystals.
Bajzíková K. et al. J. Mater. Chem. C, 2016, 4, 11540-11547. https://doi.org/10.1039/C6TC04346A
Chiral liquid crystals
The effect of the length of terminal n-alkyl carboxylate chain on selfassembling and photosensitive properties of chiral lactic acid derivatives
A new series of photosensitive azo materials possessing a chiral alkyl lactate moiety and terminal n-alkyl carboxylate unit close to the azo group has been synthesized and studied. The length of the n-alkyl carboxylate chain has been systematically varied in order to establish the effect of the molecular structure on the self-assembling behaviour. Two series of materials possessing hexyl and dodecyl alkyl chains in the chiral part of the molecule have been studied. It has been shown that the length of both the alkyl chains strongly influences the mesomorphic behaviour, however, each chiral/achiral chain has different utility to tune the mesomorphic properties. With exception of the compound with the longest chains, all studied compounds exhibited the chiral tilted ferroelectric smectic C* phase. Based on the combination of terminal alkyl chains, chiral nematic, orthogonal smectic A*, and twist grain boundary smectic A* phases have been detected on cooling beyond the SmC* phase. The presence of the photosensitive functional N=N group in the molecular core allowed further tuning of the material properties by UV light illumination. The E-to-Z photoisomerization of the azo group and subsequent thermal back-isomerisation have been studied in solution by nuclear magnetic resonance and most importantly in the mesophases on bulk samples. We report on UV-induced isothermal switching from chiral smectic and nematic mesophases into the isotropic phase, respectively, and differences in the textures of mesophase upon restoration of the ordered liquid.
Poryvai A. et al. J. Mol. Liq. 2019, 275, 829-838. https://doi.org/10.1016/j.molliq.2018.11.058
Novel chiral ion-exchangers
Strong cation exchange-type chiral stationary phase for enantioseparation of chiral amines in subcritical fluid chromatography
A new strong cation exchange type chiral stationary phase (SCX CSP) based on a syringic acid amide derivative of trans-(R, R)-2-aminocyclohexanesulfonic acid was applied to subcritical fluid chromatography (SFC) for separation of various chiral basic drugs and their analogues. Mobile phase systems consisting of aliphatic alcohols as polar modifiers and a broad range of amines with different substitution patterns and lipophilicity were employed to evaluate the impact on the SFC retention and selectivity characteristics. The observed results point to the existence of carbonic and carbamic acid salts formed as a consequence of reactions occurring between carbon dioxide, the alcoholic modifiers and the amine species present in the sub/supercritical fluid medium, respectively. Evidence is provided that these species are essential for affecting ion exchange between the strongly acidic chiral selector units and the basic analytes, following the well-established stoichiometric displacement mechanisms. Specific trends were observed when different types of amines were used as basic additives. While ammonia gave rise to the formation of the most strongly eluting carbonic and carbamic salt species, simple tertiary amines consistently provided superior levels of enantioselectivity. Furthermore, trends in the chiral SFC separation characteristics were investigated by the systematic variation of the modifier content and temperature. Different effects of additives are interpreted in terms of changes in the relative concentration of the transient ionic species contributing to analyte elution, with ammonia-derived carbamic salts being depleted at elevated temperatures by decomposition. Additionally, in an effort to optimize SFC enantiomer separation conditions for selected analytes, the impact of the type of the organic modifier, temperature, flow rate and active back pressure were also investigated.
Wolrab D. et al. J. Chromatogr. A 2013, 1289, 94-104. https://doi.org/10.1016/j.chroma.2013.03.018
Chiral separation
Consequences of transition from liquid chromatography to supercritical fluid chromatography on the overall performance of a chiral zwitterionic ion-exchanger
Major differences in the chromatographic performance of a zwitterion ion-exchange type (ZWIX) chiral stationary phase (CSP) in supercritical fluid chromatography (SFC) and high-performance liquid chromatography (HPLC) have been observed. To explain these differences, transition from HPLC to SFC conditions has been performed. The amount of a protic organic modifier in supercritical carbon dioxide (scCO2) was stepwise increased and the effect of this change studied using acidic, basic and ampholytic analytes. At the same time, the effect of various basic additives to the mobile phase and transient acidic buffer species, formed by the reaction of scCO2 with the organic modifier and additives, was assessed. Evidence is provided that a transient acid together with the intrinsic counter-ions present in the ZWIX selector structure drive the elution of analytes even when no buffer is employed. We show that the tested analytes can be enantioseparated under both SFC and HPLC conditions; the best conditions for the resolution of ampholytes are in the so-called enhanced-fluidity mobile phase region. As a consequence, subcritical fluid and enhanced-fluidity mobile phase regions seem to be chromatographic modes with a high potential for operating ZWIX CSPs.
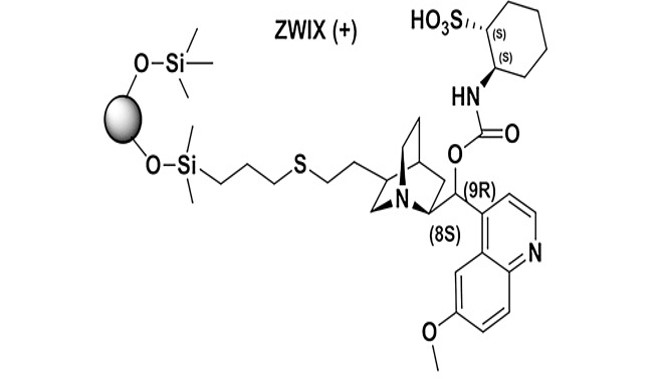
Wolrab D. et al. J. Chromatogr. A 2017, 1517, 165-175. https://doi.org/10.1016/j.chroma.2017.08.022
Biologically active substances
Synthesis, absolute configuration and in vitro cytotoxicity of deschloroketamine enantiomers: rediscovered and abused dissociative anaesthetic
In this study, we aim to determine differences in cytotoxicity of racemic deschloroketamine and its enantiomers. The synthesized racemate of this recently rediscovered and abused dissociative anaesthetic was resolved by chiral HPLC and the absolute configuration of the enantiomers was assigned using a combination of circular dichroism methods and single-crystal X-ray. Not only the absolute configuration, but also the most preferred conformers present in the crystal were successfully determined by electron and vibrational circular dichroism supported by ab initio calculations, and confirmed by X-ray. The in vitro cytotoxicity of racemic deschloroketamine and its enantiomers was determined for nine different types of cell lines. Generally, (S)-deschloroketamine exhibited higher cytotoxicity in the majority of cases. For human embryonic kidney cells (HEK 293T), the (S)-enantiomer reached the IC50 below 1 mM concentration and, in consequence, proved to be twice as potent as the (R)-enantiomer. On the other hand, live-cell fluorescence microscopy imaging at sub-IC50 concentrations provided evidence for only a minor effect of deschloroketamine racemate and enantiomers on endoplasmic reticulum stress and mitochondria morphology in neuroblastoma cells SH-SY5Y.
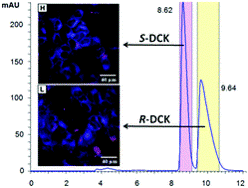
Jurásek B. et al. New J. Chem., 2018, 42, 19360-19368. https://doi.org/10.1039/C8NJ03107J
Continuous flow organocatalysis
Silica gel-immobilized multidisciplinary materials applicable in stereoselective organocatalysis and HPLC separation
In this pilot study, we present novel bifunctional silica gel-immobilized materials applicable as heterogeneous organocatalysts and stationary phases in HPLC. The materials provided high stereoselectivity in both batch and continuous flow catalysis of a model Michael addition (cyclohexanone to (E)-β-nitrostyrene). In the batch reaction, the catalysts proved their sustainable catalytic activity over five consecutive recycling experiments. Under continuous flow reaction conditions, the catalytic activity was found to be superior to the batch reaction, and moreover, the same immobilized materials were utilized as stationary phases in HPLC showing very good chemoselective separation of model acidic analytes.
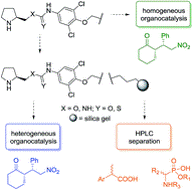
Tůma J. et al. RSC Adv., 2018, 8, 1174-1181. https://doi.org/10.1039/C7RA12658A
[urlnadstranka] => [obrazek] => [pozadi] => [poduzel] => stdClass Object ( [52380] => stdClass Object ( [nazev] => Kohout's Group [barva_pozadi] => cervena [uslideru] => false [text] => [iduzel] => 52380 [canonical_url] => [skupina_www] => Array ( ) [url] => [sablona] => stdClass Object ( [class] => infobox [html] => [css] => [js] => [autonomni] => 0 ) ) ) [iduzel] => 52382 [canonical_url] => [skupina_www] => Array ( ) [url] => /research-groups/kohout-en/research [sablona] => stdClass Object ( [class] => stranka [html] => [css] => [js] => [autonomni] => 1 ) ) [52383] => stdClass Object ( [nazev] => [seo_title] => Publications [seo_desc] => [autor] => [autor_email] => [obsah] =>Patent
Gaálová, F. Yalcinkaya, P. Cuřínová, M. Kohout, I. Stibor, P. Izák: Kompozitní chirální membrána, způsob její přípravy a způsob obohacování směsí enantiomerů, číslo patentu: 308513, datum udělení: 2.9.2020.
Publications 2023
- Jurásek, B.; Fagan, P.; Dolenský, B.; Paškanová, N.; Dobšíková, K.; Raich, I.; Jurok, R.; Setnička, V.; Kohout, M.; Čejka, J.; Kuchař, M. A structural spectroscopic study of dissociative anaesthetic methoxphenidine. New J. Chem. 2023, 47, 4543.
https://doi.org/10.1039/d2nj06126k - Dobšíková, K.; Javorská, Ž.; Paškan, M.; Spálovská, D.; Trembuláková, P.; Herciková, J.; Kuchař, M.; Kozmík, V.; Kohout, M.; Setnička, V. Enantioseparation and a comprehensive spectroscopic analysis of novel synthetic cathinones laterally substituted with a trifluoromethyl group. Spectrochim. Acta, Part A, 2023, 291, 122320.
https://doi.org/10.1016/j.saa.2023.122320
Publications 2022
- Paškan, M.; Rimpelová, S.; Svobodová Pavlíčková, V.; Spálovská, D.; Setnička, V.; Kuchař, M.; Kohout, M.: 4-Isobutylmethcathinone—A novel synthetic cathinone with high in vitro cytotoxicity and strong receptor binding preference of enantiomers. Pharmaceuticals 2022, 15, 1495.
https://doi.org/10.3390/ph15121495 - Hao, L.; Jing, H.; Xiang, Y.; Iljin, A.; Wang, Y.; Li, H.; Li, Q.; Peng, J.; Kohout, M.: Transient optically induced grating and underlying transport process in bent-core nematics. J. Appl. Phys. 2022, 132, 065108.
https://doi.org/10.1063/5.0096106 - Sysel, P.; Hovorka, Š.; Kohout, M.; Holakovský, R.; Žádný, J.; Čížek, J.; Izák, P. Optically active polyimides with different thermal histories of their preparation. Chirality 2022, 34, 1151-1161.
https://doi.org/10.1002/chir.23476 - Asnin, L.; Herciková, J.; Lindner, W.; Klimova, Y.; Ziganshina, D.; Reshetova, E.; Kohout, M.: Chiral separation of dipeptides on Cinchona-based zwitterionic chiral stationary phases under buffer-free reversed-phase conditions. Chirality 2022, 34, 1065-1077.
https://doi.org/10.1002/chir.23471 - Malinčík, J.; Kohout, M.; Svoboda, J.; Stulov, S.; Pociecha, D.; Böhmová, Z.; Novotná, V. Photochromic spiropyran-based liquid crystals. J. Mol. Liq. 2022, 346, 117842.
https://doi.org/10.1016/j.molliq.2021.117842
Publications 2021
- Kohout, M.;J. Mol. Liq. 2021, 339, 116744 DOI:10.1016/j.molliq.2021.116744
- Skopalová H., Kozmík V., Šmahel M., Svoboda J., Pacherová O., Kohout M. Mesomorphic properties of non-symmetric bent-core liquid crystals with a lateral substituent in the apex position. Liq. Cryst. 2021, 48, 1010-1024
https://doi.org/10.1080/02678292.2020.1836567 - Cigl M., Hampl F., Svoboda J., Podoliak N., Stulov S., Kohout M., Novotná V. Laterally substituted biphenyl benzoates – synthesis and mesomorphic properties. Liq. Cryst. 2021, 48, 526-536
https://doi.org/10.1080/02678292.2020.1794069 - Spálovská D., Paškan M., Jurásek B., Kuchař M., Kohout M., Setnička V. Structural spectroscopic study of enantiomerically pure synthetic cathinones and their major metabolites, New J. Chem. 2021, 45, 850.
https://doi.org/10.1039/D0NJ05065B - Wolrab D., Frühauf P., Kolderová N., Kohout M. Strong cation- and zwitterion-exchange-type mixed-mode stationary phases for separation of pharmaceuticals and biogenic amines in different chromatographic modes. J. Chromatogr. A 2021, 1635, 461751.
https://doi.org/10.1016/j.chroma.2020.461751 - Jágerová, D.; Šmahel, M.; Poryvai, A.; Macháček, J.; Novotná, V.; Kohout, M.: Photosensitive bent-core liquid crystals with laterally substituted azobenzene unit. Crystals, 2021, 11, 1265.
https://doi.org/10.3390/cryst11101265 - Karongo, R.; Ge, M.; Horak, J.; Gross, H.; Kohout, M.; Lindner, W.; Lämmerhofer, M.: Rapid enantioselective amino acid analysis by ultra-high performance liquid chromatography-mass spectrometry combining 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate derivatization with core-shell quinine carbamate anion exchanger separation. J. Chromatogr. Open, 2021, 1, 100004.
https://doi.org/10.1016/j.jcoa.2021.100004 - Herciková, J.; Spálovská, D.; Frühauf, P.; Izák, P.; Lindner, W.; Kohout, M.: Design and synthesis of naphthalene-based chiral strong cations exchangers and their application for chiral separation of basic drugs. J. Sep. Sci., 2021, 44, 3348-3356. DOI: 10.1002/jssc.202100127
Publications 2020
- Poryvai A., Bubnov A., Kohout M. Chiral Photoresponsive Liquid Crystalline Materials Derived from Cyanoazobenzene Central Core: Effect of UV Light Illumination on Mesomorphic Behavior. Crystals 2020, 10, 1161.
https://doi.org/10.3390/cryst10121161 - Tlustý M., Spálovská D., Kohout M., Eigner V., Lhoták P. Ketone transformation as a pathway to inherently chiral rigidified calix[4]arenes. Chem. Commun. 2020, 56, 12773.
https://doi.org/10.1039/D0CC05352J - Skopalová H., Špaček P., Kozmík V., Svoboda J., Novotná V., Pociecha D., Kohout M. The Role of Substitution in the Apex Position of the Bent-Core on Mesomorphic Properties of New Series of Liquid Crystalline Materials. Crystals, 2020, 10, 735. https://doi.org/10.3390/cryst10090735
- Guselnikova O., Postnikov P., Kolská Z., Záruba K., Kohout M., Elashnikov R., Švorčík V., Lyutakov O. Homochiral metal-organic frameworks functionalized SERS substrate for atto-molar enantio-selective detection. Appl. Mater. Today 2020, 20, 100666.
https://doi.org/10.1016/j.apmt.2020.100666 - Kolderová N., Jurásek B., Kuchař M., Lindner W., Kohout M. Gradient supercritical fluid chromatography coupled to mass spectrometry with a gradient flow of make-up solvent for enantioseparation of cathinones. J. Chromatogr. A 2020, 1625, 461286. https://doi.org/10.1016/j.chroma.2020.461286
- Tlustý M., Dvořáková H., Čejka J., Kohout M., Lhoták P. Regioselective Formation of Quinazoline Moiety on the Upper Rim of Calix[4]arene as the Way to Inherently Chiral Systems. New J. Chem. 2020, 44, 6490.
https://doi.org/10.1039/D0NJ01035A - Šmahel M., Poryvai A., Xiang Y., Pociecha D., Troha T., Novotná V., Svoboda J., Kohout M. Photosensitive bent-core nematic liquid crystals with various linking units in the side arms: Structure-properties relationships. J. Mol. Liq. 2020, 306, 112743. https://doi.org/10.1016/j.molliq.2020.112743
- Cigl M., Jurok R., Hampl F., Svoboda J., Podoliak N., Novotná V.: Lateral substituted phenyl biphenylcarboxylates ‒ non-chiral analogues of ferroelectric liquid crystals. Liq. Cryst. 2020, 47, 768-776.
https://doi.org/10.1080/02678292.2019.1679903 - Gaálová J., Yalcinkaya F., Cuřínová P., Kohout M., Yalcinkaya B., Koštejn M., Jirsák J., Stibor I., Bara J. E., Van der Bruggen B., Izák P. Separation of racemic compound by nanofibrous composite membranes with chiral selector. J. Membr. Sci. 2020, 596, 117728.
https://doi.org/10.1016/j.memsci.2019.117728 - Otmar M., Gaálová J., Žitka J., Brožová L., Cuřínová P., Kohout M., Hovorka Š., Bara J. E., Van der Bruggen B., Jirsák J., Izák P. Preparation of PSEBS membranes bearing (S)-(-)-methylbenzylamine as chiral selector. Eur. Polym. J. 2020, 122, 109381. https://doi.org/10.1016/j.eurpolymj.2019.109381
Publications 2019
- Jing, H.; Xu, M.; Xiang, Y.; Wang, E.; Liu, D.; Poryvai, A.; Kohout, M.; Éber, N.; Buka, A.: Light tunable gratings based on flexoelectric effect in photoresponsive bent-core nematics. Adv. Opt. Mater. 2019, 1801790.
https://doi.org/10.1002/adom.201801790 - Pokluda, A.; Kohout, M.; Chudoba, J.; Krupička, M.; Cibulka, R.: Nitrosobenzene: Reagent for the Mitsunobu esterification reaction. ACS Omega 2019, 4, 5012-5018.
https://doi.org/10.1021/acsomega.8b03551 - Spálovská, D.; Maříková, T.; Kohout, M.; Králík, F.; Kuchař, M.; Setnička, V.: Methylone and pentylone: Structural analysis of new psychoactive substances. Forensic Toxicol. 2019, 37, 366-377.
https://doi.org/10.1007/s11419-019-00468-z - Poryvai, A.; Bubnov, A.; Pociecha, D.; Svoboda, J.; Kohout, M.: The effect of the length of terminal n-alkyl carboxylate chain of self-assembling and photosensitive properties of chiral lactic acid derivatives. J. Mol. Liq. 2019, 275, 829-838.
https://doi.org/10.1016/j.molliq.2018.11.058 - Geibel, C.; Dittrich, K.; Woiwode, U.; Kohout, M.; Zhang, T.; Lindner, W.; Lämmerhofer, M.: Evaluation of superficially porous particle based zwitterionic chiral ion exchangers against fully porous particle benchmarks for enantioselective ultra-high performance liquid chromatography. J. Chromatogr. A 2019, 1603, 130-140.
https://doi.org/10.1016/j.chroma.2019.06.026 - Poryvai, A.; Vojtylová-Jurkovičová, T.; Šmahel, M.; Kolderová, N.; Tomášková, P.; Sýkora, D.; Kohout, M.: Determination of optical purity of lactic acid-based chiral liquid crystals and corresponding building blocks by chiral high-performance liquid chromatography and supercritical fluid chromatography. Molecules 2019, 24, 1099.
10.3390/molecules24061099 - Tlustý, M.; Eigner, V.; Babor, M.; Kohout, M.; Lhoták, P.: Synthesis of upper rim-double-bridged calix[4]arenes bearing seven membered rings and related compounds. RSC Adv. 2019, 9, 22017-22030.
https://doi.org/10.1039/C9RA05075B - Horčic, M.; Svoboda, J.; Novotná, V.; Pociecha, D.: W-shaped mesogens and variations of their molecular structure. Liq. Cryst. 2019, 46, 816-824.
https://doi.org/10.1080/02678292.2019.1573328 - Cigl, M.; Jurok, R.; Hampl, F.; Svoboda, J.; Podoliak, N.; Novotná, V.: Lateral substituted phenyl biphenylcarboxylates – non-chiral analogues of ferroelectric liquid crystals. Liq. Cryst. 2019.
https://doi.org/10.1080/02678292.2019.1679903 - Kohout, M.; Hovorka, Š.; Herciková, J.; Wilk, M.; Sysel, P.; Izák, P.; Bartůněk, V.; von Baeckmann, C.; Pícha, J.; Frühauf, P.: Evaluation of silica from different vendors as the solid support of anion-exchange chiral stationary phases by means of preferential sorption and liquid chromatography. J. Sep. Sci. 2019, 42, 3653-3661.
https://doi.org/10.1002/jssc.201900731
Publications 2018
- Jurásek, B.; Králík, F.; Rimpelová S.; Čejka, J.; Setnička, V.; Ruml, T.; Kuchař, M.; Kohout, M.:Synthesis, Absolute Configuration and in vitroCytotoxicity of Deschloroketamine Enantiomers: Rediscovered and Abused Dissociative Anaesthetic
J. Chem. 2018, 42, 19360-19368 DOI: 10.1039/c8nj03107j - Landovsky, T.; Dvorakova, H.; Eigner, V.; Babor, M.; Krupicka, M.; Kohout, M.; Lhotak, P.:Chemoselective Oxidation of Phenoxathiin-based Thiacalix[4]arene and the Stereoselective Alkylation of Products
New J. Chem.2018, 42, 20074-20086 DOI: 1039/C8NJ04690E - Schmitt, K.; Woiwode, U.; Kohout, M.; Zhang, T.; Lindner, W.; Lämmerhofer, M.:
Comparison of small size fully porous particles and superficially porous particles of chiral anion-exchange type stationary phases in ultra-high performance liquid chromatography: effect of particle and pore size on chromatographic efficiency and kinetic performance
Chromatogr. A2018, 1569, 149-159 DOI:10.1016/j.chroma.2018.07.056 - Spálovská, D.; Kralik, F.; Kohout, M.; Jurasek, B.; Habartová, L.; Kuchar, M.; Setnicka, V.: Structure determination of butylone as a new psychoactive substance using chiroptical and vibrational spectroscopies
Chirality2018, 30, 548-559 DOI:1002/chir.22825 - Wolrab, D.; Kohout, M.: Multimodální stacionární fáze pro kapalinovou chromatografii, způsob jejich přípravy a jejich použití. Národní patent č. 307339, číslo přihlášky 2017-193, datum udělení 2.5.2018, datum zveřejnění 13.6.2018.
- Kohout, M.; Wernisch, S.; Tuma, J.; Hettegger, H.; Picha, J.; Lindner W.: Effect of different immobilization strategies on chiral recognition properties of Cinchona-based anion exchangers
Sep. Sci.2018, 41, 1355-1364 DOI:10.1002/jssc.201701213 - Bajtai, A.; Fekete, B.; Palkó, M.; Fülöp, F.; Lindner, W.; Kohout, M.; Ilisz, I.; Péter, A.: Comparative study on the liquid chromatographic enantioseparation of cyclic β-amino acids and the related cyclic β-aminohydroxamic acids on Cinchona alkaloid-based zwitterionic chiral stationary phases
Sep. Sci.2018, 41, 1216-1223 DOI:10.1002/jssc.201701190 - Sardella, R.; Macchiarulo, A.; Urbinati, F.; Ianni, F.; Carotti, A.; Kohout, M.; Lindner, W.; Péter, A.; Ilisz, I.: Exploring the enantiorecognition mechanism of Cinchona alkaloid-based zwitterionic chiral stationary phases and the basic trans-paroxetine enantiomers
Sep. Sci.2018, 41, 1199-1207 DOI:10.1002/jssc.201701068 - Tuma, J.; Kohout, M.: Silica gel-immobilized multidisciplinary materials applicable in stereoselective organocatalysis and HPLC separation
RSC Adv.2018, 8, 1174-1181 DOI:1039/c7ra12658a - Kozmik, V.; Rodinová, E.; Prausová, T.; Svoboda, J.; Novotná, V.; Pociecha, D.: Mesogens with central naphthalene core substituted at various positions
Cryst. 2018, 45, 744-756 DOI: 10.1080/02678292.2017.1380238 - Bajzíková, K.; Vesely, J.; Kozmik, V.; Svoboda, J.; Novotná, V.; Pociecha, D.: Thiophene central core for the design of bent-shaped liquid crystals
Mol. Liq. 2018, 267, 496-503 DOI:10.1016/j.molliq.2018.02.009 - Horcic, M.; Svoboda, J.; Novotná, V.; Pociecha, D.; Gorecka, E.: Bent-core dimers with top-to-bottom linkage between central units
RSC Adv. 2018, 8, 22974-22985 DOI: 1039/c8ra04108c - Marz, M.; Kohout, M.; Nevesely, T.; Chudoba, J.; Prukala, D.; Nizinski, S.; Sikorski, M.; Burdzinski, G.; Cibulka, R.:Azodicarboxylate-free esterification with triphenylphosphine mediated by flavin and visible light: method development and stereoselectivity control
Biomol. Chem.2018, 16, 6809-6817 DOI:10.1039/C8OB01822G
Publications 2017
- Maixner, J.; Jurásek, B.; Kohout, M.; Kuchar, M.; Kacer, P.: X-ray powder diffraction data for (S)-Deschloroketamine hydrochloride, C13H18 Powder Diffr.2017, 32, 193-195. DOI:10.1017/S0885715617000586
- Bajzíková, K.; Svoboda, J.; Novotná, V.; Pociecha, D.; Gorecka E.: Bent-core mesogens with an aromatic unit at the terminal position. New J. Chem.2017, 41, 4672-4679 DOI: 1039/C6NJ03908A
- Žurek, J.; Svobodová, E.; Šturala, J.; Dvořáková, H.;Svoboda, J.; Cibulka, R.: Chiral ethylene-bridged flavinium salts: The stereoselectivity of flavin-10a-hydroperoxide formation and the effect of substitution on the photochemical properties. Tetrahedron Asymmetry 2017, 28, 1780-1791. DOI:1016/j.tetasy.2017.10.029
- Dobrovolny, K.; Ulbrich, P.; Svecova, M.; Rimpelova, S.; Malincik, J.; Kohout, M.; Svoboda, J.; Bartunek, V.: Copper nanoparticles in glycerol-polyvinyl alcohol matrix: In situ preparation, stabilization and antimicrobial aktivity. Alloy. Compd.2017, 697, 147-155 DOI:10.1016/j.jallcom.2016.12.144
- Kolderova, N.; Nevesely, T.; Sturala, J.; Kuchar, M.; Holakovsky, R.; Kohout, M.:
Enantioseparation of chiral sulfoxides on amylose-based columns: comparison of normal phase liquid chromatography and supercritical fluid chromatography Chromatographia 2017, 80, 547-557 DOI: 10.1007/s10337-016-3234-6 - Kohout, M.; Alaasar, M.; Poryvai, A.; Novotna, V.; Poppe, S.; Tschierske, C.; Svoboda, J.: Photosensitive bent-core liquid crystals based on methyl substituted 3-hydroxybenzoic acid
RSC Adv.2017, 7, 35805-35813 DOI:10.1039/C7RA05632J - Wolrab, D.; Fruehauf, P.; Gerner, C.; Kohout, M.;Lindner, W.: Consequences of transition from liquid chromatography to supercritical fluid chromatography on the overal performance of a chiral zwitterionic ion-exchanger. J. Chromatogr. A 2017, 1517, 165-175 DOI:10.1016/j.chroma.2017.08.022
- Pallova, L.;Kozmik, V.; Kohout, M.; Svoboda, J.; Novotna, V.; Pociecha, D.: Bent-core liquid crystals with a 2-substituted 3-hydroxybenzoic acid central core. Cryst. 2017, 44, 1306-1315 DOI: 10.1080/02678292.2016.1276981
- Tlusty, M.; Slavik, P.; Kohout, M.; Eigner, V.; Lhotak, P.: Inherently Chiral Upper-Rim-Bridged Calix[4]arenes Possessing a Seven Membered Ring
Org. Lett. 2017, 19, 2933-2936 DOI:10.1021/acs.orglett.7b01170 - Miksatko, J.; Eigner, V.; Kohout, M.; Lhotak, P.: Regio-/stereoselective Formation of Monosulfoxides from Thiacalix[4]arenes in All Possible Conformations
Tetrahedron Letters 2017, 58, 1687–1691 DOI:10.1016/j.tetlet.2017.03.043 - Horčic, M.; Kohout, M.; Svoboda, J.; Novotna, V.; Pociecha, D.; Gorecka, E.: Core-to-core Dimers Forming Switchable Mesophase
Chem. Commun. 2017, 53, 2721-2724 DOI:10.1039/C6CC09983A - März, M.; Chudoba, J.; Kohout, M.; Cibulka, R.: Photocatalytic Esterification under Mitsunobu Reaction Conditions Mediated by Flavin and Visible Light
Org. Biomol. Chem. 2017, 15, 1970-1975 DOI:10.1039/C6OB02770A
Publications 2016
- Bajzikova, K.; Kohout, M.; Tarabek, J.; Svoboda, J.; Novotna, V.; Vejpravova, J.; Pociecha, D.; Gorecka, E.: All-organic liquid crystalline radicals with a spin unit in the outer position of a bent-core systém. J. Mater. Chem. C2016, 4, 11540-11547. DOI:10.1039/C6TC04346A
- Tůma, J.; Kohout, M.; Svoboda, J.; Novotná, V.; Pociecha, D.: Bent-core liquid crystals based on 6-substituted 3-hydroxybenzoic acid: the role of substitution and linkage group orientation on mesomorphic properties. Liq. Cryst.2016, 43, 1889-1900. DOI:10.1080/02678292.2016.1230789
- Kohout, M.; Vandenbussche, J.; Roller, A.; Tůma, J.; Boqaerts, J.; Bultinck, P.; Herrebout, W.; Lindner, W.: Absolute configuration of the antimalarial erythro-mefloquine - vibrational dichroism and X-ray diffraction studies of mefloquine and its thiourea derivative. RSC Advances2016, 6, 81461-81465. DOI:1039/C6RA19367F
- Kohout, M.; Bubnov, A.; Šťurala, J.; Novotná, V.; Svoboda, J.: Effect on alkyl chain length in the terminal ester group on mesomorphic properties of new chiral lactic acid derivatives. Cryst.2016, 43, 1472-1485. DOI:10.1080/02678292.2016.1185170
- Grecsó, N.; Kohout, M.; Carotti, A.; Sardella, R.; Natalini, B.; Fülöp, F.; Lindner, W.; Péter, A.; Ilisz, I.: Mechanistic considerations of enantiorecognition on novel Cinchona alkaloid-based zwitterionic chiral stationary phases from the aspect of the separation of trans-paroxetine enantiomers as model compounds. Pharm. Biomed. Anal.2016, 124, 164-173. DOI:10.1016/j.jpba.2016.02.043
- Wolrab, D.; Frühauf, P.; Moulisova, A.; Kuchar, M.; Gerner, C.; Lindner, W.; Kohout, M.: Chiral separation of new designer drugs (Cathinones) on chiral ion-exchange type stationary phases. Pharm. Biomed. Anal.2016, 120, 306-315. DOI:10.1016/j.jpba.2015.12.023
- Tůma, J.; Kohout, M.; Svoboda, J.; Novotná, V.; Pociecha, D.: Bent-shaped liquid crystals based on 4-substituted 3-hydroxybenzoic acid central core - Part II. Cryst. 2016, 43, 547-563 DOI: 10.1080/02678292.2015.1125535
- Slavik, P.; Kohout, M.; Böhm, S.; Eigner, V.; Lhotak, P.: Synthesis of Inherently Chiral Calixarenes via Direct Mercuration of the Partial Cone Conformation. Chem. Comm. 2016, 52, 2366-2360 DOI:10.1039/C5CC09388K
Publications 2015
- Trišović, N.; Antanasijević, J.; Tóth-Katona, T.; Kohout, M.; Salamonczyk, M.; Sprunt, S.; Jákli, A.; Fodor-Csorba, K.: Azo-containing asymmetric bent-core liquid crystals with modulated smectic phase RSC Adv. 2015, 5, 64886-64891. DOI: 10.1039/c5ra09764a
- Kohout, M.; Bielec, B.; Steindl, P.; Trettenhahn, G.; Lindner, W.: Mechanistic aspects of the direct C-acylation of cyclic 1,3-diones with various unactivated carboxylic acids Tetrahedron 2015, 71, 2698-2707. DOI: tet.2015.03.037
- von Koschitzky, I.; Gerhardt, H.; Lämmerhofer, M.; Kohout, M.; Gehringer, M.; Laufer, S.; Pink, M.; Schmitz-Spanke, S.; Strube, C.; Kaiser, A.: New insights into novel inhibitors against deoxyhypusine hydrolase from plasmodium falciparum: compounds with an iron chelating potential Amino Acids 2015, 47, 1155-1166. DOI: 1007/s00726-015-1943-z
- Kohout, M.; Kozmík, V.; Slabochová, M.; Tůma, J.; Svoboda, J.; Novotná, V.; Pociecha, D.: Bent-shaped liquid crystals based on 4-substituted 3-hydroxybenzoic acid central core Cryst. 2015, 42, 87-103. DOI: 10.1080/02678292.2014.965232
- Kozmík, V.; Poznik, M.; Svoboda, J.; Frere, P.:
Dithieno[3,2-b:2',3'-d]furan as a new building block for fused conjugated systems
Tetrahedron Lett.2015, 56, 6251-6253. DOI:10.1016/j.tetlet.2015.09.107